The structures of individual molecules in crude oil remain unknown in spite of the considerable amount of research dedicated to this topic. The extreme complexity of crude oil makes it difficult to impossible to separate crude oil into individual compounds and decide their structure by NMR or X-ray spectroscopy.
In a new study, scientists reported a new approach to oil composition analysis using high temperature and pressure to dissolve crude oil in water. The new strategy is agreeable with green chemistry standards as it obviates the requirement for naturally dangerous solvents.
The extreme complexity of crude oil makes it difficult to impossible into individual compounds. Heavier fractions, which are nonvolatile at 300 degrees Celsius, are yet to be appropriately considered. It is realized that they comprise predominantly of phenols, ketones, carbazoles, pyridines, quinolines, dibenzofurans, and carboxylic acids.
In addition, crude oil from certain fields may likewise contain sulphuric compounds. Many hydrocarbons have identical formulas, with a similar number of carbon, hydrogen, and oxygen atoms, however, vary in their arrangements, i.e., they are isomers.
Different structures indeed have different chemical properties. Heavier hydrocarbons consist of many atoms, which means more structural variability for each compound.
Mass spectrometry provides information on the elemental composition of substances and their molecular mass but often fails to distinguish between different isomers. Such information may be obtained through isotope exchange analysis. This method is based on the fact that, depending on which particular compounds constitute crude oil or some other sample, oxygen and hydrogen atoms will take more or less time to be replaced with their isotopes—essentially the same elements, but with a different mass.
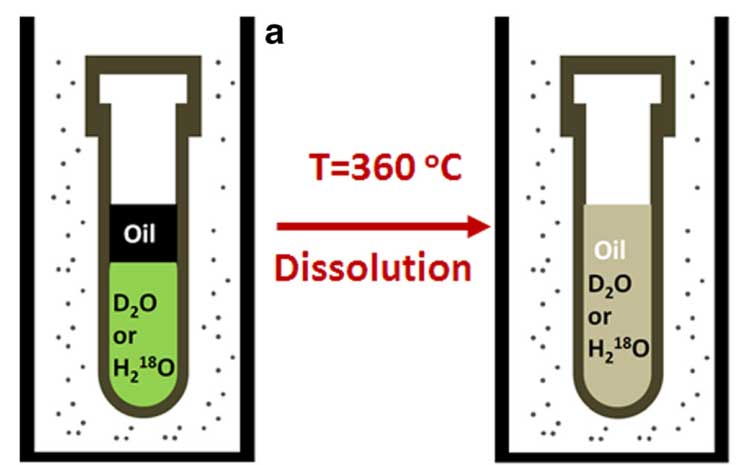
Water is the most readily available and the cleanest source of isotopes, but oil is insoluble in water under normal conditions, so potent acids and alkali have to be used instead. But acids tend to break down organic compounds, especially at high temperatures, thus altering the sample’s composition.
It is known, however, that compounds insoluble in water may be dissolved in superheated, or supercritical, water at temperatures significantly over 100 degrees C, so it was decided to apply this method to crude oil.
Scientists proved it by obtaining a water-based crude oil solution by increasing the temperature and pressure and analyzed its composition.
They heated the sample to 360 degrees C in heavy water at a pressure above 300 atm for one hour.
Scientists then compared mass spectra of the original sample and the sample after the isotope exchange reaction and obtained more information on the structure of the compounds comprising crude oil.
Professor of Skoltech and MIPT Eugene Nikolaev, who also heads the Laboratory of Mass Spectrometry at Skoltech said, “Isotope labels may only be incorporated at specific positions in the molecule, similar to the lock-and-key model. We can analyze molecular structure by using high-resolution mass spectrometry to measure the exchange rate even when it is impossible to separate individual compounds and identify their structure with other methods.”
Yury Kostyukevich, one of the authors of the paper and a senior researcher at Skoltech and MIPT laboratories said, “Light crude oil reserves are depleting. Hydrocracking of fuel oil, which is characterized by its highly complex and poorly studied molecular structure, is playing an ever greater role in gasoline production. Hydrocrackers are expensive, they are not produced in Russia, and they require the use of special catalysts. We have found a way to identify furans, pyridines, and naphthenic acids in crude oil without having to resort to the complex distillation process.”
“We hope our research will help better understand crude oil structure and composition, contribute to the development of new catalysts for more efficient oil refining, and enable improved oil quality monitoring in trunk pipeline systems.”
The study was conducted in collaboration by the MIPT, Skoltech, the Joint Institute for High Temperatures of the Russian Academy of Sciences, and Lomonosov Moscow State University. It is published in the Analytical and Bioanalytical Chemistry.
