In order to effectively address global warming, scientists across the globe are finding ways to reduce CO2. Scientists at Tokyo Tech have proposed an innovative way to achieve carbon capture using a rhenium-based electrocatalytic system that is capable of reducing low-concentration CO2 (even 1%).
Scientists drew on decades of work sharpening the capacities of a rhenium-based catalyst and showed its capacity to reduce low-fixation CO2 within the sight of a concoction called triethanolamine (TEOA).
Contrasted with numerous past examinations that have concentrated on decreasing unadulterated CO2, few have investigated how to enhance coordinate catch of low-concentration CO2 — a point that warrants further examination, taking into account that plants outfit low convergences of CO2 (about 400ppm, that is 0.04% of the climate) and exhaust gases from overwhelming enterprises commonly contain low levels of CO2 (around 3– 13%).
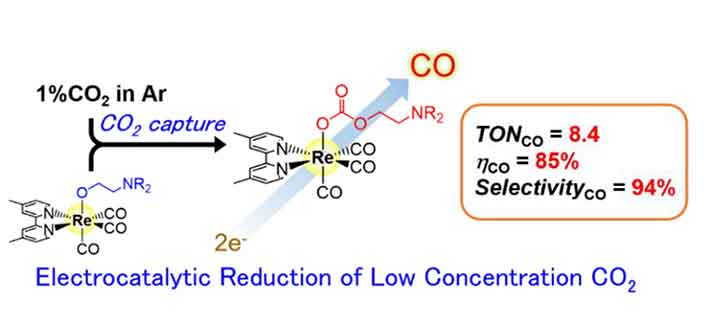
Electrocatalytic reduction of low-concentration CO2 was achieved using a rhenium-based complex with high CO2-capturing ability.
By dodging the requirement for extra vitality expending buildup forms, their strategy, whenever scaled up, could give a more suitable, ecologically well-disposed answer for a CO2 catch in numerous settings.
In a progression of investigations to survey electrocatalytic action, the analysts found that at a CO2 grouping of 1%, the rhenium-based impetus demonstrated high selectivity (94%) towards carbon monoxide (CO) development.
Scientists noted, “A likely reason behind the high performance is the efficient insertion of CO2 into the rhenium–oxygen bond.”
Now, scientists want to continue systematically investigating promising strategies to help reduce real-world CO2 levels.
The study is published in the journal Chemical Science.
