Proteins and molecules assemble and disassemble as a major aspect of numerous fundamental organic procedures. It is exceptionally hard to watch these components, which are frequently mind-boggling and happen at the nanometer scale. EPFL scientists now discovered a way that allows these mechanisms to be examined with unprecedented precision.
To observe processes on a sample that changes over time, the scanning speed has to be increased. This new method controls the physical interaction of the sharp tip very precisely using pulsed laser light. This dramatically increased the scanning speed while preserving the gentle but extremely precise scanning motion.
Prof. Georg Fantner, who leads EPFL’s Laboratory for Bio- and Nano- Instrumentation (LBNI) said, “Change is essential for living matter and is therefore crucial to biological processes. So it was essential that we find a way to observe it.”
Adrian Nievergelt, a PhD student at LBNI and co-first author of the paper said, “We achieved this by using two lasers in the microscope, one of which is pointed at the base of the cantilever, locally heating it and thereby bending it slightly. By bending the cantilever, we can probe the surface much quicker, while still keeping fine control of the overall movement. In addition, we improved the overall system’s performance, allowing us to scan up to 2,000 lines per second.”
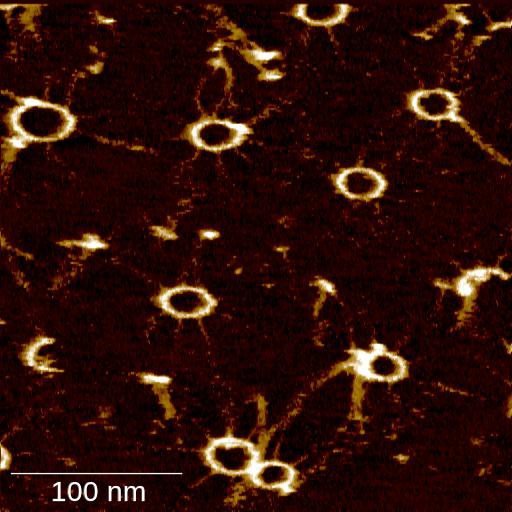
The researchers tested this new technology to analyze the dynamics of SAS-6 protein ring formation. This protein family plays a key role in the assembly of centrioles, which are minute organelles conserved from algae to men, fundamental for cell motility and division. Scientists found that the method allows them to visualize the various stages of the ring assembly of SAS-6 proteins in real time.
Prof. Pierre Gönczy, an expert in centriole biology said, “This is a critical game changer for the field.
Niccolò Banterle, a post-doctoral fellow in the Gönczy laboratory and co-first author of the study said, “Now we finally have a method to directly observe how this critical cellular component is assembled into a ring-like polymer. This allows us to better understand how nature controls the assembly of some of the smallest building blocks of life.”
Their work is the subject of a paper published in Nature Nanotechnology.
