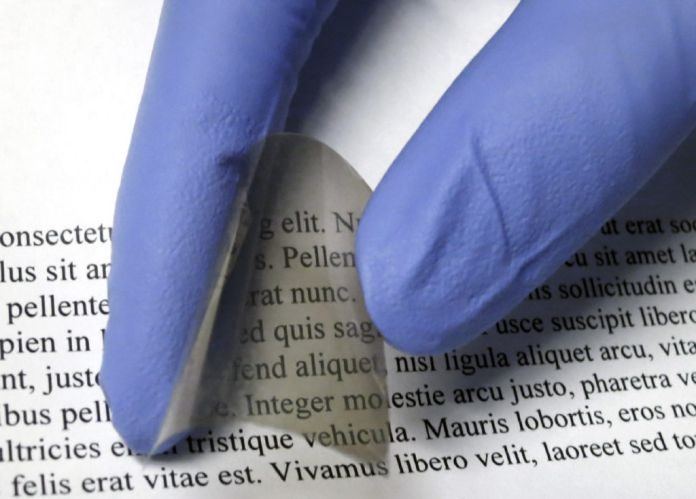
Scientists from Cockrell School of Engineering at the University of Texas at Austin have developed a novel smart windows material. They have developed a flexible Electrochromic device, which has an amorphous structure and passes small electric charge of 4 volts. The device can lighten or darken the material and control the transmission of heat-producing, near-infrared radiation. Such smart windows are aimed at saving on cooling and heating bills for homes and businesses.
Scientists have advanced a new low-temperature process. Through this process, the material is covered in plastic easily and cheaply. Thus, windows, sunroofs or curved glass surfaces has the potential to control heat and light when both are integrated with this material. The process also creates a material with a decent nanostructure. Thus, it doubles the effectiveness of the coloration process as compared to coating produced by a complex high-temperature process. By using less power, it switches between clear and shaded instantly.
Although, the new process produce a different local arrangement of the atoms in a sequence, chain-like structure. This new linearly structured material, made of chemically abbreviated niobium oxide. It allows ions to flow in and out independently. It becomes twice in energy effectiveness as the conventionally processed smart window material.
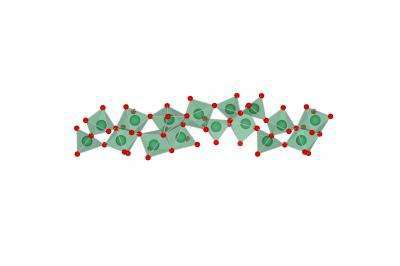
During research, Scientists vision’s was in the atomic-scale structure of the amorphous materials. There were some messy structures, which are hard to analyze. There are only a few techniques available to examine atomic-scale structure sufficiently with its properties. But still, it is difficult to engineer amorphous materials with improved performance.
Delia Milliron said, “There’s relatively little insight into amorphous materials and how their properties are impacts by the local structure. But, we characterized with enough specificity what the local arrangement of the atoms is. Thus, it sheds light on the differences in properties in a rational way.” (Milliron is an associate professor in the McKetta Department of Chemical Engineering.)
Graeme Henkelman, a chemistry professor in UT Austin’s College of Natural Sciences, explains, “determining the atomic structure of amorphous materials is far more difficult than for crystalline materials, which have an ordered structure. In this case, the researchers used a combination of techniques and measurements to determine an atomic structure that is consistent in both experiment and theory. Such collaborative efforts that combine complementary techniques are, in my view, the key to the rational design of new materials.”
According to scientists, the research will inspire deliberate engineering of amorphous materials for other applications. For example, supercapacitors that store and release electrical energy instantly and effectively.
Scientists are now trying to create a flexible material using their low-temperature process.The material will meet or exceeds the best performance of electrochromic materials made by conventional high-temperature processing.