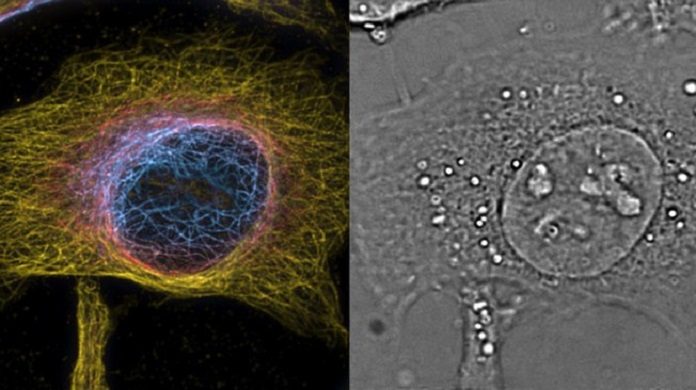
EPFL scientists have developed a new super-resolution technique that can perform spatial and temporal imaging, capturing unprecedented views inside living cells. The technique holds the potential to see beyond the diffraction of light, providing unprecedented views of cells and their interior structures and organelles.
Since the technique won the Nobel Prize in Chemistry in 2014, it has gathered interest among developers for various applications. But it has a big limitation: it only offers a spatial resolution.
Scientists now have created steps to address the issue by building up a strategy that can perform both 3D super-resolution microscopy and quick 3D phase imaging in a solitary instrument. Phase imaging is a procedure that interprets the adjustments in the period of light caused by cells and their organelles into refractive list maps of the cells themselves.
Scientists dubbed the technique as a 4D microscope that combines the sensitivity and high time-resolution of phase imaging with the specificity and high spatial resolution of fluorescence microscopy. The researchers developed a novel algorithm that can recover the phase information from a stack of bright-field images taken by a classical microscope.
Adrien Descloux, one of the lead authors of the paper said, “With this algorithm, we present a new way to achieve 3D quantitative phase microscopy using a conventional bright-field microscope. This allows direct visualization and analysis of subcellular structures in living cells without labeling.”
The technique involves custom designing of an image-splitting prism, which allows the simultaneous recording of a stack of eight z-displaced images. Though this, the microscope can perform high-speed 3D phase imaging across a volume of 2.5μm x 50μm x 50μm.
Kristin Grussmayer, another one of the paper’s lead authors said, “With the prism as an add-on, you can turn a classical microscope into an ultra-fast 3D imager. It is also suited for 3D fluorescence imaging, which the scientists tested using super-resolution optical fluctuation imaging (SOFI) that exploits the blinking of fluorescent dyes to improve 3D resolution through correlation analysis of the signal. Using this, the researchers performed 3D super-resolution imaging of stained structures in the cells, and combined it with 3D label-free phase imaging.”
Professor Hilal Lashuel said, “We are thrilled by these results and the possibilities offered by this technique. The technical advances enabled high-resolution visualization of the formation of pathological alpha-synuclein aggregates in hippocampal neurons.”
Theo Lasser said, “We offer PRISM as a new microscopy tool and anticipate that it will be rapidly used in the life science community to expand the scope of 3D high-speed imaging for biological investigations. We hope that it will become a regular workhorse for neuroscience and biology.”
The landmark paper is published in Nature Photonics.