Pancreatic islets can offer the details on the normal glucose level in different species. Also, the first programming stays, notwithstanding when the islets are transplanted starting with one species then onto the next. For instance, pancreatic islets are transplanted from humans to mice, they continue to dictate the blood glucose levels that are normal for humans.
This suggests that islets mimic as glucostat to maintain to keeps glucose levels within a normal range for any given species.
A new study by the University of Miami Miller School of Medicine offers insights on cellular functions that can go awry in people living with diabetes. According to scientists, this finding could have important implications for treatment and research.
Conducting the study on mice, scientists transplanted pancreatic islets from different species into diabetic and non-diabetic mice. They then measured glycemia and glucose tolerance in the recipient mice.
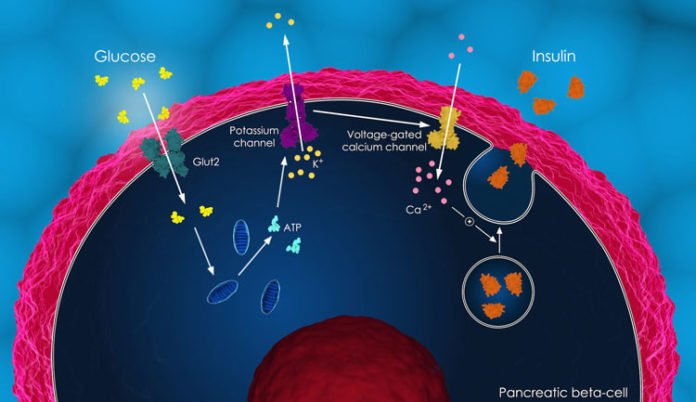
A second vital revelation is that the beta cells in the pancreas — the main cells in the human body that make and discharge insulin — don’t act alone. Other endocrine cells in the pancreatic islet are a piece of a complex creation that assist the human body to maintain its “glycemic set point” — or normal blood glucose level.
Rayner Rodriguez-Diaz, Ph.D., research assistant professor of medicine said, “The islet imposes its glycemic set point on the organism, making it the bona fide glucostat in the body.”
“These findings impact transplantation and regenerative approaches to treat diabetes. Furthermore, therapeutic strategies using glucagon receptor antagonists to reduce high blood glucose in diabetics need to be reassessed, as they may negatively impact the function of the cells that produce insulin, the hormone diabetic patients need the most.”
Scientists demonstrated that mediations that exclusive target beta cells are in this way likely missing the full picture. In addition, scientists discovered that unlike with rodent islets, glucagon input from the alpha cell to the insulin-secreting beta cell is necessary to fine-tune the distinctive human set point.
Scientists are further planning to continue studying the complex physiology of the human islet in vivo in mice. Collaborating with investigators at Columbia University in New York, for example, they plan to induce and follow the maturation of stem cell-derived human pseudo-islets in vivo.
Miller School co-authors of the study were Alejandro Caicedo, Ph.D., Ruth Molano, D.V.M., Jonathan Weitz, Ph.D., Midhat H. Abdulreda, Ph.D., Dora M. Berman, Ph.D., Norma Sue Kenyon, Ph.D., Camillo Ricordi, M.D., Antonello Pileggi, M.D., Ph.D. Research collaborators from the Karolinska Institutet in Stockholm were Barbara Leibiger, Ingo B. Leibiger, Ph.D., and Per-Olof Berggren, Ph.D.
Their findings were published in the journal Cell Metabolism.