A metal is a material that, when freshly prepared, polished, or fractured, shows a lustrous appearance, and conducts electricity and heat relatively well.
But how, exactly, is a metallic conduction band formed from localized electrons initially, and what is the corresponding microscopic picture for the material involved?
In a new study, a collaboration of scientists from the Czech Republic, the U.S., and Germany, the research team of Pavel Jungwirth from the Institute of Organic Chemistry and Biochemistry of the Czech Academy of Sciences (IOCB Prague) has found the answer to this question.
Scientists have mapped the electrolyte-to-metal transition in alkali metal—liquid ammonia solutions using a combination of photoelectron spectroscopy (PES) and electronic structure calculations at the molecular level.
Alkali metals dissolve in liquid ammonia represent archetypal systems to explore the change from blue electrolytes at low concentrations to bronze-or gold-colored metallic solutions (with conductivity practically identical to a copper wire) with higher concentrations of excess electrons.
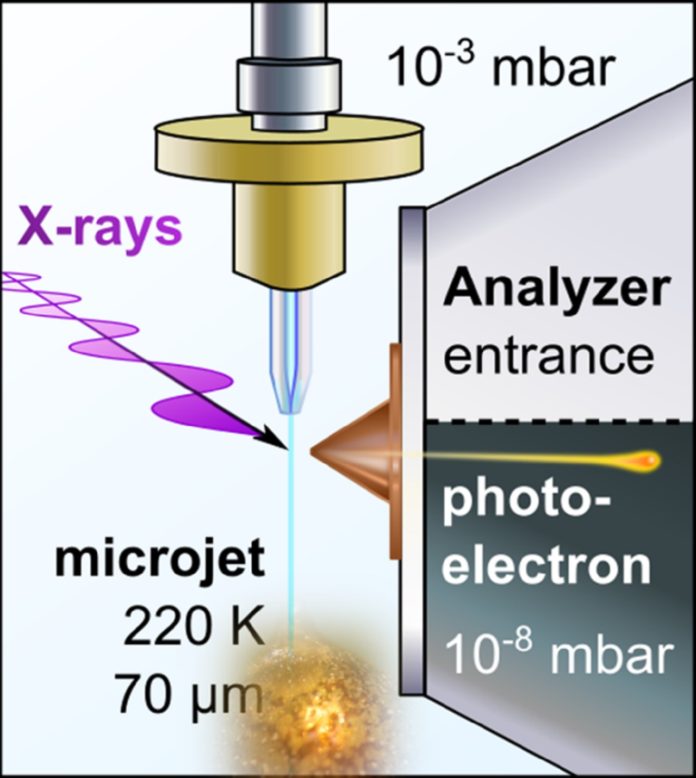
Simultaneously, PES represents an entire apparatus for building up the electronic structure relevant to this change. As an ultra-high vacuum method, PES was long thought to be incompatible with volatile liquids until the technique of liquid microjets was developed for water and aqueous solutions.
In 2019, the group of Pavel Jungwirth, in collaboration with colleagues at the University of Southern California and the BESSY II synchrotron in Berlin, performed the first successful PES measurements on a refrigerated polar liquid—pure liquid ammonia.
This achievement paves the way towards PES studies of alkali metal—liquid ammonia systems, which map the electrolyte-to-metal transition for lithium, sodium, and potassium dissolved in liquid ammonia using PES using soft X-ray synchrotron radiation.
Hence, scientists captured for the first time the photoelectron signal of excess electrons in liquid ammonia as a peak at around 2 eV binding energy. This peak then broadens asymmetrically toward higher binding energies upon increasing the alkali metal concentration, gradually forming a conduction band with a sharp Fermi edge accompanied by plasmon peaks, both of which are fingerprints of the nascent metallic behavior.
Along with the electronic structure calculations, these measurements offer a detailed molecular picture of the transition from a non-metal to a metal. This enables scientists to better comprehend the onset of metallic behavior characterized by properties such as high electric conductivity.
Pavel Jungwirth said, “Hopefully, the present work on metallic ammonia will open the path to realizing our most ‘explosive’ idea: the preparation of metallic water by very carefully mixing it with alkali metals.”
Journal Reference:
- Tillmann Buttersack et al. Photoelectron spectra of alkali metal–ammonia microjets: From blue electrolyte to bronze metal. DOI: 10.1126/science.aaz7607