Inflammatory bowel disease (IBD) represents a group of intestinal disorders that cause prolonged inflammation of the digestive tract. Patients usually suffer from pain, extreme discomfort, and numerous other symptoms caused by continuously relapsing and remitting inflammatory lesions in the layer of cells that lines the intestinal lumen (mucosa).
The nature and cause of changes in the microbiota in patients with IBD are poorly understood. Yet, it is clear that a misdirected immune system is at work, and that certain components of the microbial community in our gut, known as the intestinal microbiome, and environmental factors contribute to its destructive forces.
Currently, there are anti-inflammatory drugs available commercially to dampen acute inflammation and antibiotics to fight local infections when IBD episodes flare-up. However, they are expensive. Intriguingly, these drugs can have severe side effects, and antibiotics can disrupt the beneficial parts of the microbiome on which we depend on many of our body’s functions.
Importantly, there are no wound treatments available that could be applied to inflamed lesions directly from inside the gut lumen to speed up the healing process and minimize the use of those drugs.
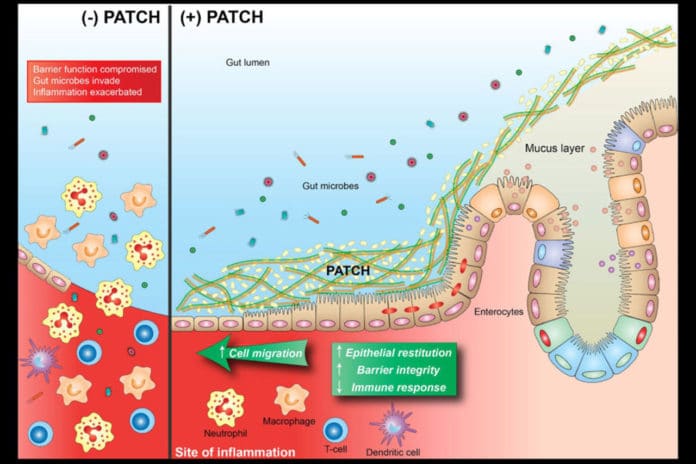
Now, scientists from Harvard’s Wyss Institute for Biologically Inspired Engineering have developed a living-material approach that uses a strain of genetically engineered E.coli Nissle gut bacteria as a locally acting probiotic. The bacteria generate a network of nanofibers that directly binds to mucus to fill inflamed areas like a patch, shielding them from gut microbes and environmental factors.
Tested on mice, this strategy was able to protect mice against the effects of colitis induced by a chemical agent and promoted mucosal healing.
Neel S. Joshi, Ph.D., from Wyss Institute said, “With this ‘living therapeutics’ approach, we created multivalent biomaterials that are secreted by resident engineered bacteria on-site and attach to many mucus proteins at a time — firmly adhering to the viscous and otherwise moving mucus layer, which is a challenging thing to do.”
“The Probiotic Associated Therapeutic Curli Hybrids (PATCH) approach, as we named it, creates a biocompatible, mucoadhesive coating that functions as a stable, self-regenerating Band-Aid and provides biological cues for mucosal healing.”
In this study, scientists introduced the machinery that produces one of the mucoadhesive hydrogels based on TFF3 into an E. coli Nissle strain that is a normal gut bacterium that can thrive in the colon and cecum sections of the intestinal tract affected by IBD and is currently sold in many commercial probiotic formulations.
First author Pichet Praveschotinunt, who is a graduate student mentored by Joshi, said, “We found that the newly engineered Nissle bacteria, when given orally, also populated and resided in the intestinal tract, and that their curli fibers integrated with the intestinal mucus layer.”
“When we induced colitis in the colons of mice by orally administering the chemical dextran sodium sulfate, animals that had received the PATCH-generating E. coli Nissle strain by daily rectal administration starting three days prior to chemical treatment had significantly faster healing and lower inflammatory responses, which caused them to lose much less weight and recover faster compared to control animals. Their colon epithelial mucosa displayed more normal morphology and lower numbers of infiltrating immune cells.”
Scientists believe that their approach can be used as a companion therapy to existing anti-inflammatory, immuno-suppressant, and antibiotic therapies to help minimize patients’ exposure to the drugs and potentially provide protection against IBD relapses.
Their findings are reported in Nature Communications.
Additional authors on the study are Wyss Institute researchers Ilia Gelfat, Franziska Bahl, and David B. Chou.
The study was supported by a grant from the National Institutes of Health, funds from Harvard’s Wyss Institute for Biologically Inspired Engineering and the Blavatnik Biomedical Accelerator, and a royal Thai government scholarship.