Ceramic fuel cells are a kind of high-temperature fuel cell that operates over 800. This high temperature permits the utilization of inexpensive catalysts, nickel, as opposed to low-temperature fuel cells, for example, polymer electrolyte fuel cells, which utilize expensive platinum catalysts to supplement their low catalytic activity.
In any case, amusingly, even though high-temperature fuel cells use inexpensive catalysts, their activity requires costly refractory materials and manufacturing technologies. Another constraining factor is that their on-off system procedure takes a long time because of the qualities of high-temperature operation, which limit their application to large stationary power generation systems.
Scientists around the world have tried to develop thin-film-based ceramic fuel cells, which can operate at low temperatures without performance loss.
Tragically, the issue is that lower-temperature operation makes ceramic fuel cells lose one of their significant advantages, that is, their capacity to use different fuels. At the point when the nickel catalyst of the fuel cell is utilized with hydrocarbon fuels, for example, methane, propane, and butane, the carbon produced during fuel change are stored on the outside of nickel. This worsens severely with lower temperatures, prompting the failure of the cell operation.
Now, scientists at the Korea Institute of Science and Technology addressed this problem. They have developed a high-performance ceramic fuel cell that can operate on butane fuels.
Scientists have developed a high-performance, thin-film-based ceramic fuel cell that could operate at mid-to-low temperatures below 600 °C using butane fuels.
As butane can be liquified, thus this new technology is expected to expand the application range of ceramic fuel cells to portable and mobile applications such as electric cars, robots, and drones.
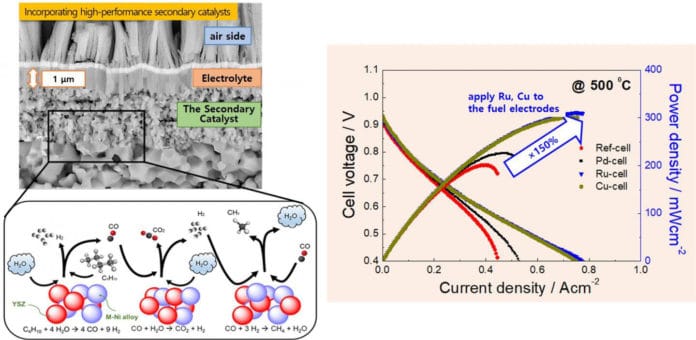
Specifically, scientists used alternating deposition of the secondary catalyst, and the primary catalyst layers to efficiently disperse the secondary catalyst at the earliest parts of the fuel electrodes to the electrolyte.
In this way, controlled incorporation of small amounts yet adequately situated secondary catalysts was conceivable.
Through this process, scientists successfully applied secondary catalysts known for their high catalytic activity at low temperatures, such as palladium (Pd), ruthenium (Ru), and copper (Cu), to the nano-structure fuel electrodes.
They confirmed the high-performance operation of the newly developed thin-film-based ceramic fuel cells at mid and low operation temperatures (500-600 ?), using butane fuel, which is a very affordable fuel.
Dr. Son Ji-Won at KIST’s Center for Energy Materials Research, said, “This research systematically examined the possible uses of hydrocarbon fuels in ceramic fuel cells operating at low temperatures. The use of portable fuels like butane at lower operating temperatures would enable the development of smaller and integrated ceramic fuel cell systems, which can be applied to portable and mobile power sources.”
Journal Reference:
- Effect of secondary metal catalysts on butane internal steam reforming operation of thin-film solid oxide fuel cells at 500–600 °C. DOI: 10.1016/j.apcatb.2019.118349