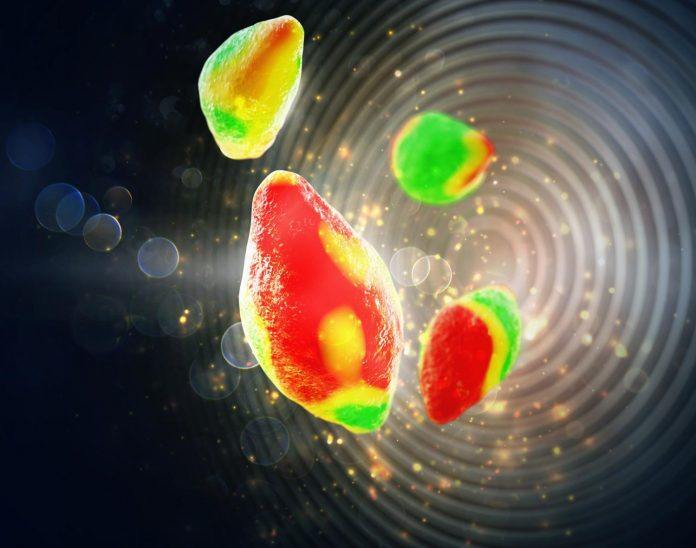
Scientists from Stanford University have recently devised a novel technique to reveal nanoscale secrets of lithium ion batteries. This novel method is X-Ray microscopy technique, which peers as never before into the electrochemical reaction that fuels the batteries.
Scientists developed this complex method by picturing the basic building block of batteries. This X-Ray microscopy technique allows researchers to observe and understand the electrochemistry of battery.
Jongwoo Lim, said, “The method gives basic vision into battery working. Previously, various studies were invented to observe the behavior of battery. Now, we can see and understand how individual battery particles charge and discharge.” (Jongwoo Lim is a co-lead author of the paper and post-doctoral researcher at the Stanford Institute for Materials & Energy Sciences at SLAC).
The basic process of charging and discharging are restricted by an electrochemical Achilles heel. Ions uniformly inserts across particle’s surface. Instead, certain areas take on more ions, and others fewer. These inconsistencies eventually lead to mechanical stress as areas of the crystal lattice become overburdened with ions and develop tiny fractures, sapping battery performance and shortening battery life.
According to Yiyang Li, a doctoral candidate in Chueh’s lab, “Charging and discharging process should be uniform and homogeneous. But hey are non- uniform in reality. In our better understanding of the process, this paper lays out a path toward suppressing the phenomenon.”

Improving the batteries is like debating these damaging forces. This may lead to batteries that charge faster and more fully, lasting much longer than today’s models.
Martin Bazant said, “The surprising phenomenon reveled by this X-Ray microscopy technique, is game changing in battery field.” (Martin Bazant is a professor of chemical engineering and of mathematics at MIT who led the theoretical aspect of the study.)
Scientists then developed a transparent battery with same active materials. It contains two very thin, transparent silicon nitride windows. The battery electrode was made by single lithium iron phosphate nanoparticle’s layer, which sits on membrane inside the gap between two windows. A salty fluid, known as an electrolyte, flows in the gap to deliver the lithium ions to the nanoparticles.
William Chueh said, “This was a extremely small battery, holding ten billion times less charge than a smartphone battery. But it allows us a clear view of what’s happening at the nanoscale.” (William Chueh, an assistant professor of materials science and engineering at Stanford and a faculty scientist at the Department of Energy’s SLAC National Accelerator Laboratory)
In research, scientists found, charging process is essentially less uniform than discharging process. They also found that faster charging improves uniformity, which causes new and better battery designs and power management strategies. Improved uniformity improves battery cyclability and reduce damaging mechanical stress on the electrodes.
“Beyond batteries, this work could have a far-reaching impact on many other electrochemical materials. It may be used in catalysts, memory devices, and so-called smart glass,” said Chueh.
We learned that here is not just how to make a better battery, but offers us a profound new window on the science of electrochemical reactions at the nanoscale,” Bazant said.