The Pauli exclusion principle suggests that no two fermions can have the same set of quantum numbers. It also indicates that no two fermions can occupy the same quantum state.
Meanwhile, electrons must have different orbits around a nucleus, and by extension, it explains why atoms have volume. This comprehension of self-ordering of fermions has prompted different discoveries—for example, that they should form crystals with a particular geometry, which are currently known as Pauli crystals. When this observation was first made, it was comprehended that such crystal formation could occur under specific conditions.
In this new study, the specialists have settled the circumstances and, in this manner, have constructed an apparatus that allowed them to observe Pauli crystals for the first time.
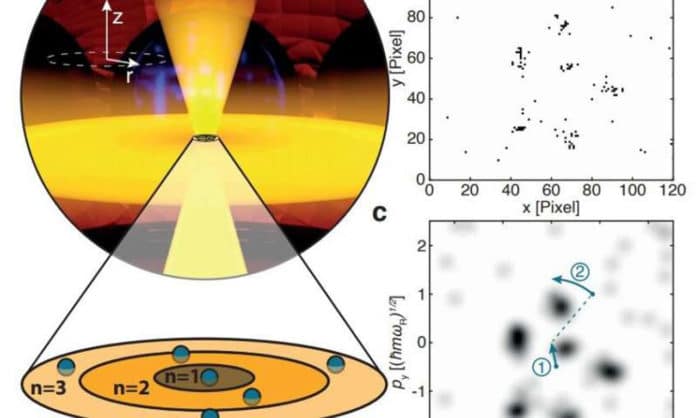
Scientists at Heidelberg University conducted this study. During the study, that involved a setup with lasers to trap a cloud of lithium-6 atoms supercooled to their lower energy state, forcing them to adhere to the exclusion principle, in a one-atom-thick flat layer.
They next utilized a strategy that permitted them to image the toms when they were in a specific given state— and only those atoms. They then used the camera to take 20,000 pictures, yet utilized just those that demonstrated the correct number of atoms— – showing that they were adhering to the Pauli exclusion principle.
Next, the team prepared the rest of the images to evacuate the effect of overall momentum in the atom cloud, pivoted them appropriately, and then superimposed thousands of them, uncovering the momentum disturbance of the individual molecules—that was where crystals started to rise in the photographs, similarly as was anticipated by hypothesis. The specialists note that their method could likewise be utilized to consider different impacts identified with fermion-based gases.
Journal Reference:
- Marvin Holten, Observation of Pauli Crystals. arXiv:2005.03929