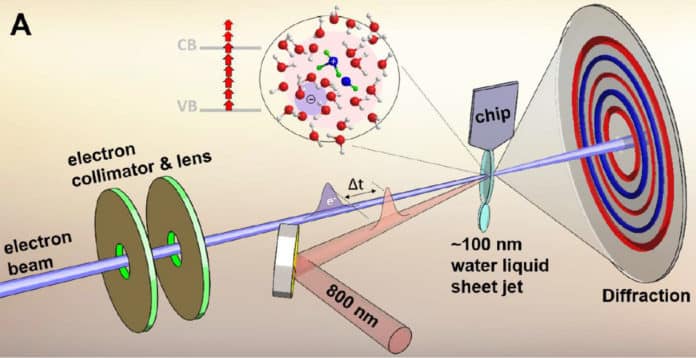
Using a high-speed electron camera called MeV-UED, scientists at the Department of Energy’s SLAC National Accelerator Laboratory have unveiled vital steps in the ionization of liquid water.
Interaction of high-energy radiation with water molecules triggers a series of ultrafast reactions. At first, it expels an electron and keeps a positively charged water molecule. This process happens so fast- within a fraction of a trillionth of a second where the water molecule gives up a proton to another water molecule.
This creates a hydroxyl radical (OH) – which can damage virtually any macromolecule in an organism, including DNA, RNA, and proteins—and a hydronium ion (H3O+), which are abundant in the interstellar medium and tails of comets, and might contain clues about the origin of life.
In a previous study using SLAC’s Linac Coherent Light Source (LCLS) X-ray laser, scientists witnessed the ultrafast proton transfer reaction following ionization of liquid water. But until now, scientists have yet to observe the hydroxyl-hydronium pair directly.
SLAC scientist and study lead Ming-Fu Lin said, “All laser surgeries and radiotherapies produce this unstable complex, which may lead to many chemical reactions in the human body. Interestingly, this complex also helps to purify our drinking water by killing germs. It is also of importance in nuclear power generation where other forms of radiation ionize water. Many simulations predict the existence of this complex, but now we have finally observed its formation.”
To see the short-lived hydroxyl-hydronium pair, scientists created 100-nanometer-thick jets of liquid water. They ionized the water molecules with intense laser light.
Then they probed the molecules with short pulses of high-energy electrons from MeV-UED to generate high-resolution snapshots of the ionization process. This allowed them to measure bonds between oxygen atoms and bonds between oxygen and hydrogen atoms simultaneously, thus capturing this important but unstable complex.
Scientists are further planning to increase the imaging speed so the proton transfer process can be measured directly before forming the hydroxyl-hydronium pairs.
Matthias Ihme, an associate professor in the Stanford University Mechanical Engineering Department who led the theoretical analysis, said, “Simulations have intensively studied both topics, but no direct structural measurements have been taken to validate theories. These measurements are also critical for testing our theoretical models that predict these processes.”
Xijie Wang, a SLAC distinguished staff scientist and study collaborator, said, “Many intermediate states and structures in chemical reactions are either unknown or have yet to be observed directly. We can use MeV-UED to explore and capture various short-lived and important complexes, opening a window to study chemical reactions as they occur.”
The study has fundamental significance to various fields, including nuclear engineering, space travel, cancer treatment, and environmental remediation.
Journal Reference:
- M.-F. Lin et al., Imaging the short-lived hydroxyl-hydronium pair in ionized liquid water, Science (2021). DOI: 10.1126/science.abg3091