Scientists from the Weizmann Institute of Science have- for the first time ever- grown synthetic embryo models of mice outside the womb using stem cells cultured in a petri dish. This is a major medical science breakthrough.
What’s more, the cell culture process is also done without using fertilized eggs, thereby bypassing the need for sperm.
This approach could, to a significant extent, avoid the technological and moral concerns associated with using natural embryos in research and biotechnology, making it incredibly important. Even with mice, some tests are currently impractical because they would necessitate thousands of embryos; however, the availability of models developed from mouse embryonic cells, which multiply by the millions in lab incubators, is almost unlimited.
Prof. Jacob Hanna of Weizmann’s Molecular Genetics Department said, “The embryo is the best organ-making machine and the best 3D bioprinter – we tried to emulate what it does. Scientists already know how to restore mature cells to “stemness” – pioneers of this cellular reprogramming had won a Nobel Prize in 2012. But going in the opposite direction, causing stem cells to differentiate into specialized body cells, not to mention entire form organs, has proved much more problematic.”
“Until now, in most studies, the specialized cells were often either hard to produce or aberrant, and they tended to form a mishmash instead of well-structured tissue suitable for transplantation. We managed to overcome these hurdles by unleashing the self-organization potential encoded in the stem cells.”
In March 2021, scientists came up with the electronically controlled device that allows the growing of natural mouse embryos outside the womb. Scientists used the same device in the new experiment to grow mouse stem cells for over a week – about half the gestation time for a mouse.
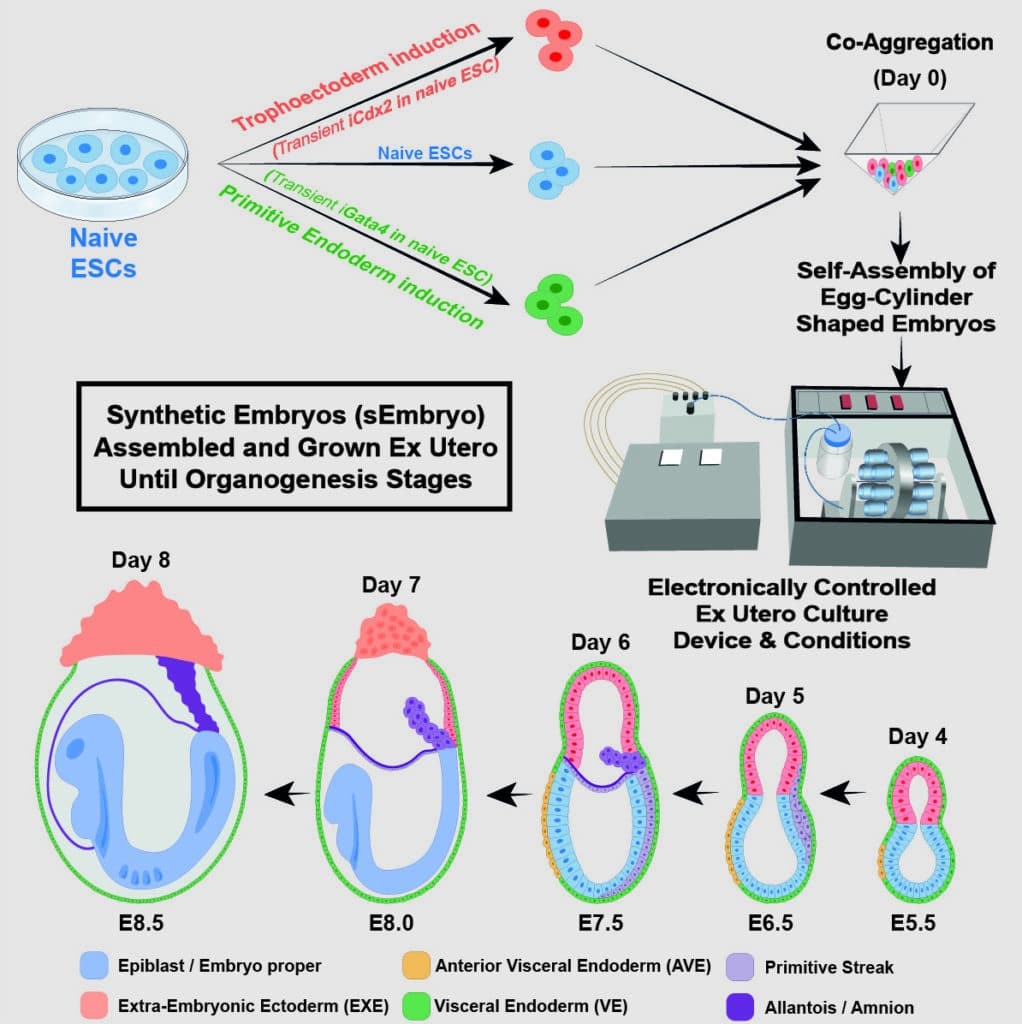
The scientists divided the stem cells into three groups before being placed into the device. In one, the cells that would eventually become embryonic organs themselves were left in their current state. One of two types of genes, master regulators of either the placenta or the yolk sac, were overexpressed in the cells in the other two groups during a pretreatment that lasted only 48 hours.
Hanna said, “We gave these two groups of cells a transient push to give rise to extraembryonic tissues that sustain the developing embryo.”
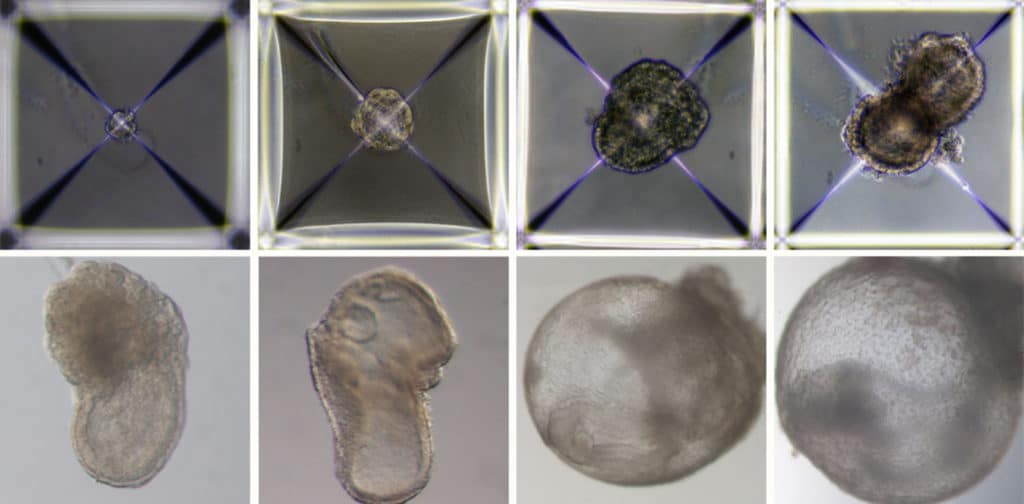
The three cell groups were quickly combined inside the device to form aggregates, the majority of which failed to undergo full development. However, 50 out of 10,000, or roughly 0.5 percent, went on to form spheres, each of which later developed into an elongated, embryo-like structure. The placenta and yolk sacs were visible outside the embryos, and the model’s development proceeded like in a natural embryo since the scientists had colored-coded each set of cells.
These synthetic models developed normally until day 8.5 – nearly half of the mouse’s 20-day gestation – at which stage all the early organ progenitors had formed, including a beating heart, blood stem cell circulation, and a brain with well-shaped folds, a neural tube, and an intestinal tract. Compared to natural mouse embryos, the synthetic models displayed a 95 percent similarity in both the shape of internal structures and the gene expression patterns of different cell types. The organs seen in the models gave every indication of being functional.
Hanna said, “The study presents a new arena: Our next challenge is understanding how stem cells know what to do – how they self-assemble into organs and find their way to their assigned spots inside an embryo. And because our system, unlike a womb, is transparent, it may prove useful for modeling birth and implantation defects of human embryos.”
“Instead of developing a different protocol for growing each cell type – for example, those of the kidney or liver – we may one day be able to create a synthetic embryo-like model and then isolate the cells we need. We won’t need to dictate to the emerging organs how they must develop. The embryo itself does this best.”
Journal Reference:
- Shadi Tarazi, Alejandro Aguilera Castrejon, et al. ¬Post-Gastrulation Synthetic Embryos Generated Ex Utero from Mouse Naïve ESCs. Cell August 01, 2022. DOI: 10.1016/j.cell.2022.07.028