Proteins- active sites of enzymes- accelerates and guide biochemical reactions make life possible. To do this, their instructions have to conflict somewhat with the protein’s overall structural plan, suggests a new study by the Rice University scientists.
Evolution has chosen parts of compounds as being imperative enough to be permitted to vivaciously digress from the guidelines that lead to a folding funnel, which manages the proteins into their functional low-vitality states. By looking over basically all known proteins, the specialists likewise affirmed the suspicion that extra dimensions of all the more feebly baffled cooperations encompass and bolster the catalytic sites themselves.
The study broadens the principle of minimal frustration that clarifies why proteins can easily advance from the direct arrangements initially encoded in their DNA to their working, three-dimensional globular structures. This principle recognizes the way that dissatisfaction frequently goes uncertainly when the staying fiery clash some way or another serves the protein’s function.
According to Rice’s Peter Wolynes, who enunciated the principle in a mathematical form more than 20 years ago, the energy landscapes of proteins with catalytic parts are worth extra analysis, given the importance evolution has assigned them.
He said, “A perfectly folded molecule, no matter how beautiful as a sculpture, can’t do much. You have to have a protein that, while being mostly folded, still has a few frustrated hinges that are incompatible with folding but are necessary to allow for the movement required for chemical reactions.”
“Frustration at the atomic level is simple to experience: Just push the positive poles of two magnets together and feel how they push back. Like magnets, the amino acids of a folding protein attract and repel each other as they (mostly) resolve conflicts and finally come to form a stable arrangement of contacts.”
“Now add a third magnet, and you can encounter frustration that you can’t get rid of.”
Frustration among the enzyme’s neighboring amino acids, as in the circumstance with the third magnet, acts to enable the frustrated enzyme to hold fast. It remains fairly insecure yet has locales accessible to tie with and modified target molecules.
Wolynes said, “Clearly, evolution has priorities that supersede the aesthetics of achieving a perfect fold.”
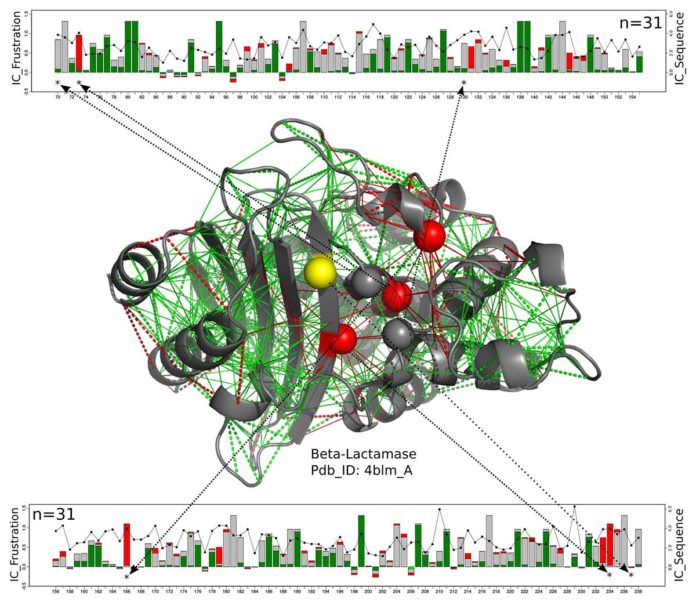
Scientists studied different types of protein structures and discovered that entire families of enzymes share characteristic signatures of active-site frustration that extend to the second and third amino acid layers, or shells, around the enzyme.
Wolynes said, “The interesting thing we found was that frustration generally extends beyond the absolute first shell. That means there’s an awkward but necessary subtlety to the functional constraints that requires getting these three shells right.”
“We have seen evidence of extended frustration in their efforts to modify enzymes for novel reactions. Sometimes they would specifically change the active site in order to catalyze different chemical reactions from the natural ones, but found that amino acids distant from the active site also needed to change to make the enzyme work efficiently.”
“It wasn’t obvious why, but the models show that changes in the second and third shells do improve enzymes’ catalytic ability. So our results were not a complete shock, but it’s nice to see that extended frustration is quite prevalent in nature and is a common theme through all the known enzyme classes.”
The study is detailed in the Proceedings of the National Academy of Sciences.