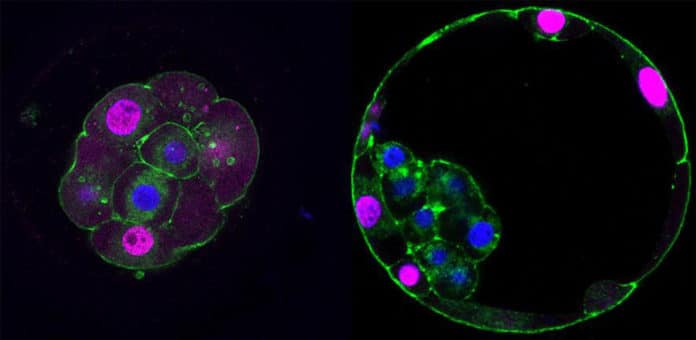
Current understandings of cell specification in early mammalian pre-implantation development are based mainly on mouse studies.
A new study by the University of Cambridge looks at biological pathways active in human embryos during their first few days of development. Through this study, scientists wanted to understand how cells acquire different fates and functions within the early embryo.
The study has shown that just after the fertilization, when cells start to divide- some cells start to stick together. This triggers a cascade of molecular events that start placental development. A subset of cells change shape, or ‘polarise,’ which drives the change into a placental progenitor cell – the precursor to a specific placenta cell – that can be recognized by contrasts in genes proteins from different cells in the embryo.
Dr. Kathy Niakan, group leader of the Human Embryo and Stem Cell Laboratory at the Francis Crick Institute, said, “This study highlights the critical importance of the placenta for healthy human development.”
“If the molecular mechanism we discovered for this first cell decision in humans is not appropriately established, this will have significant negative consequences for the development of the embryo and its ability to implant in the womb successfully.”
Scientists additionally analyzed similar developmental pathways in mouse and cow embryos. They found that while the mechanisms of later stages of development vary between species, the placental progenitor is as yet the first cell to separate.
Claudia Gerri, the lead author of the study and postdoctoral training fellow in the Human Embryo and Stem Cell Laboratory at the Francis Crick Institute, said, “We’ve shown that one of the earliest cell decisions during development is widespread in mammals, and this will help form the basis of future developmental research. Next, we must further interrogate these pathways to identify biomarkers and facilitate healthy placental development in people, and also cows or other domestic animals.”
“During IVF, one of the most significant predictors of an embryo implanting in the womb is the appearance of placental progenitor cells under the microscope. If we could identify better markers of placental health or find ways to improve it, this could make a difference for people struggling to conceive.”
Niakan said, “Understanding the process of early human development in the womb could provide us with insights that may lead to improvements in IVF success rates in the future. It could also allow us to understand early placental dysfunctions that can pose a risk to human health later in pregnancy.”
The research was led by scientists at the Francis Crick Institute in collaboration with colleagues at the Royal Veterinary College, Vrije Universiteit Brussel, Université de Nantes, and Bourn Hall Clinic. Kathy Niakan is the incoming Director of the University of Cambridge’s Centre for Trophoblast Research, and Chair of the Cambridge Strategic Research Initiative in Reproduction.
Journal Reference:
- Gerri, C. et al.: ‘Initiation of a conserved trophectoderm program in human, cow and mouse embryos.’ Nature, Sept 2020. DOI: 10.1038/s41586-020-2759-x.