UNIST scientists have developed a new technology for hydrogen production from the process, which involves an electron that is produced during the decomposition of biomass, such as waste wood. Their new system decomposes lignin with a molybdenum (Mo) catalyst to create high value-added compounds, and the electrons removed in the process adequately produce hydrogen.
Hydrogen is a promising energy vector for the future. Among the different methods of its production, the electrolysis of water has attracted great attention because it is sustainable and renewable chemical technology.
Be that as it may, in the currently reported technology, the oxygen generation reaction (OER) is sow and complicated, and hydrogen production efficiency is low. This is because hydrogen gas (H₂) is produced by hydrogen particles (H⁺) as electrons because these electrons originate from the oxygen evolution reaction.
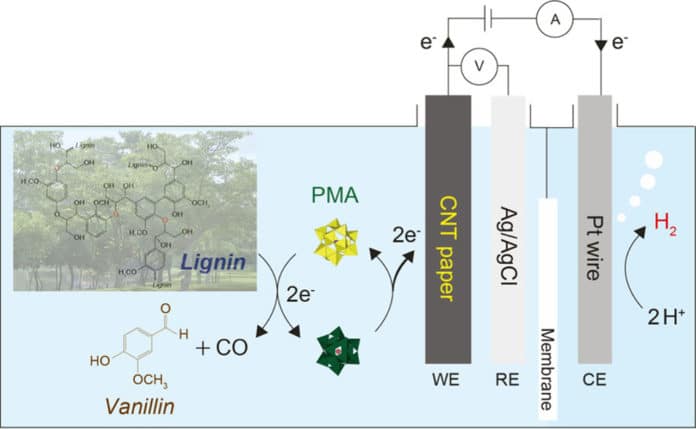
In this new system, lignin is used as an electron donor in a manner to diminish the overall inefficiency of the oxygen evolution reaction (OER). This is the principle of using molybdenum-based inexpensive metal impetuses (PMA) to break down lignin at low temperatures and concentrate the electrons created in the process to deliver hydrogen. The new device has been intended to move particles from lignin, along the wire to the electrode where the hydrogen evolution reaction (HER) occurs.
Hyeonmyeong Oh (Combined M.S/Ph.D. of Energy and Chemical Engineering, UNIST) said, “With this new system, we can produce hydrogen with less energy (overvoltage) than conventional water electrolysis, as there is no need for oxygen reactions, requiring high energy and precious metal catalysts. Conventional methods require more than 1.5 volts, but the new system was capable of producing hydrogen at a much lower potential (0.95 volts).”
Research Assistant Professor Yuri Choi, the co-author of the study, said, “Also, vanillin or carbon monoxide (CO), which are produced via lignin breakdown, is a beneficial substance for various industrial processes. Lignin, the second most naturally abundant biomass, is difficult to decompose. However, using molybdenum-based catalysts (PMA), it was easily degraded at low temperatures.”
Professor Jungki Ryu in the School of Energy and Chemical Engineering at UNIST said, “The new biofuel system is a technology that produces hydrogen and valuable chemicals using cheap catalysts and low voltages instead of expensive catalysts such as platinum (Pt). Our work is also significant, as it presents a new way to replace oxygen-producing reactions in the electrolysis of water.”
The findings of this research have been published in ACS Catalysis on January 3, 2020.