Scientists at the University of Washington in collaboration with the Department of Energy’s Pacific Northwest National Laboratory and University of California, Los Angeles- have created new design rules for engineers to create microelectronics, membranes, and tissues and open up better production methods for new materials.
Some materials required to undergo a particular rule while assembling on surfaces, one row at a time. For instance, Nucleation, the first process, should be prevalent in ordered structures across nature and technology, from cloud droplets to rock candy.
Yet despite some predictions made in the 1870s by the American scientist J. Willard Gibbs, researchers are still debating how this basic process happens.
In a new study, scientists studied Gibbs’ theory for materials that form row by row. They found the underlying mechanism, which fills in a fundamental knowledge gap and opens new pathways in materials science.
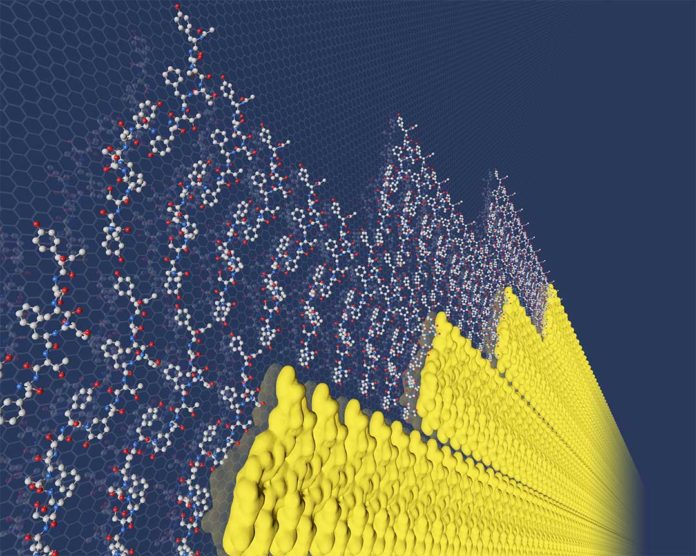
During the experiment, scientists used small protein fragments called peptides that show specificity, or unique belonging, to a material surface.
The UCLA collaborators have been identifying and using such material-specific peptides as control agents to force nanomaterials to grow into certain shapes, such as those desired in catalytic reactions or semiconductor devices. The research team made the discovery while investigating how a particular peptide — one with a strong binding affinity for molybdenum disulfide — interacts with the material.
PNNL materials scientist James De Yoreo said, “It was complete serendipity. We didn’t expect the peptides to assemble into their own highly ordered structures.”
Co-corresponding author Yu Huang said, “That may have happened because “this peptide was identified from a molecular evolution process. It appears nature does find its way to minimize energy consumption and to work wonders.”
According to Gibbs’ classical nucleation theory, although turning the water into ice saves energy, creating the solid-liquid interface costs energy. The tricky part is the initial start — that’s when the surface area of the new particle of ice is large compared to its volume, so it costs more energy to make an ice particle that is saved.
Gibbs’ theory predicts that if the materials can grow in one dimension, meaning row by row, no such energy penalty would exist. Then the materials can avoid what scientists call the nucleation barrier and are free to self-assemble.
De Yoreo said, “But this study shows there are certainly cases where Gibbs’ theory works well.”
Scientists found a way to understand how molecular interactions with materials impact their nucleation and growth. They exposed the peptide solution to fresh surfaces of a molybdenum disulfide substrate, measuring the interactions with atomic force microscopy. Then, they compared the measurements with molecular dynamics simulations.
In the early stage, they found the peptides bound to the material one row at a time, barrier-free, just as Gibbs’ theory predicts.
Using atomic force microscopy, scientists were able to see the rows while forming. The results showed the rows were ordered right from the start and grew at the same speed regardless of their size — a key piece of evidence. They also formed new rows as soon as enough peptide was in the solution for existing rows to grow; that would only happen if row formation is barrier-free.
De Yoreo said, “This row-by-row process provides clues for the design of 2D materials. Currently, to form certain shapes, designers sometimes need to put systems far out of equilibrium or balance. That is difficult to control.”
“But in 1D, the difficulty of getting things to form in an ordered structure goes away. Then you can operate right near equilibrium and still grow these structures without losing control of the system.”
Huang said, “Now with the new understanding, we can start to exploit the specific interactions between molecules and 2D materials for automatic assembly processes.”
De Yoreo said, “The next step is to make artificial molecules that have the same properties as the peptides studied in the new paper — only more robust.”
The research was published online Dec. 6 in the journal Science.