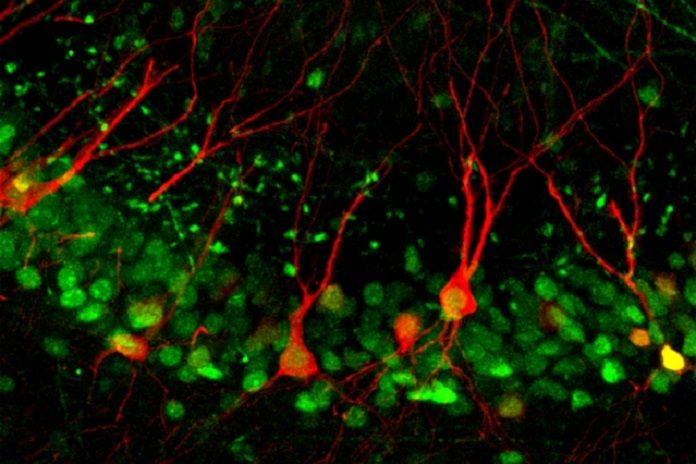
MIT neuroscientists have unveiled a cellular pathway that permits particular synapses to become stronger during memory building. The results procure the first scintilla of the molecular mechanism by which prolonged reminiscences are encoded in a region of the hippocampus called CA3.
In a study published in 2011, Lin and colleagues discovered that Npas4, a gene that is turned on instantly following novel experiences, appears to act as a chief controller of the program of gene expression needed for prolonged reminiscence building. They likewise discovered that Npas4 is the most active in the CA3 region of the hippocampus during learning.
This activity was previously known to be required for fast contextual learning same is required during a kind of task studied as contextual fear conditioning. During the conditioning, mice receive a mild electric shock when they step in and explore a specific chamber. Within minutes, the mice learn to fear the chamber, and the post they step in it, they freeze.
When the researchers knocked out the Npas4 gene, they discovered that mice could not recall the tearful episode. They likewise investigated the same effect when they knocked out the gene mere in the CA3 region of the hippocampus. Knocking it out in other parts of the hippocampus, however, had no impact on reminiscence.
In the new study, the researchers explored in more detail how Npas4 exerts its impacts.
Lin’s lab had antecedently invented a technique that makes it possible to fluorescently label CA3 neurons that are activated during this fear conditioning. Using the same fear conditioning process, the researchers demonstrated that during learning, certain synaptic inputs to CA3 neurons are strengthened, but not others. Furthermore, this strengthening requires Npas4.
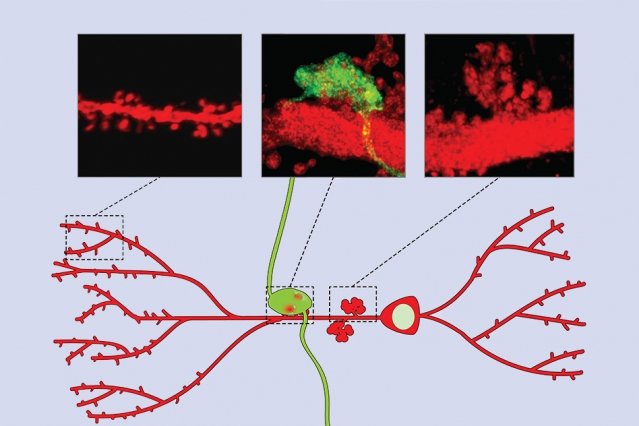
Image: Lin Lab
The inputs that are selectively strengthened received from another part of the hippocampus called the dentate gyrus. These signals convey information about the location where the fearful experience took place. Without Npas4, synapses receiving from the dentate gyrus to CA3 failed to strengthen, and the mice could not build reminiscences of the event.
Later in the experiment unveiled that this strengthening is necessary particularly for reminiscence encoding, not for retrieving reminiscences previously built.
The researchers likewise discovered that Npas4 loss did not impact synaptic inputs that CA3 neurons receive from other sources.
Kimberly Raab-Graham, an associate professor of physiology and pharmacology at Wake Forest University School of Medicine, says the researchers used an impressive variety of techniques to unequivocally show that contextual memory formation is tightly controlled by Npas4.
Raab-Graham, who was not involved in the study, stated, “The major finding of the study is that contextual memory is driven by a single circuit and comes down to a single transcription factor.”
“When they knocked out the transcription factor, they removed contextual memory formation, and they could restore it by adding the transcription factor.”
Post these scientists discovered that a protein called Npas4, antecedently recognized as a cardinal controller of gene expression prompt by neuronal activity controls the strength of connections amid neurons in the CA3 and those in another part of the hippocampus called the dentate gyrus.
In the absence of Npas4, prolonged reminiscences cannot build.
Yingxi Lin, an associate professor of brain and cognitive sciences said, “Our study identifies an experience-dependent synaptic mechanism for memory encoding in CA3, and provides the first evidence for a molecular pathway that selectively controls it.”
Neuroscientists have previously investigated that the brain encodes reminiscences by changing the strength of synapses, or connections amid neurons. This needs interactions of many proteins discovered in both presynaptic neurons, which dispatch information about an event, and postsynaptic neurons, which adopt the information.
Neurons in the CA3 region play a critical role in the building of contextual reminiscences, which are reminiscences that clinch an event with the location where it took place, or with other contextual information such as timing or emotions. These neurons adopt synaptic inputs from three distinct pathways, and scientists have hypothesized that one of these inputs, from the dentate gyrus, is critical for encoding novel contextual reminiscences. However, the mechanism of how this information is encoded was ambiguity.
The researchers further investigated one of the genes that Npas4 controls to exert this impact on synapse strength. This gene, known as plk2, is involved in shrinking postsynaptic structures. Npas4 turns on plk2, thereby reducing synapse size and strength. This suggests that Npas4 itself does not strengthen synapses, but maintains synapses in a state that permits them to be strengthened when required. In absence of Npas4, synapses become too strong and therefore cannot be induced to encode memories by further strengthening them.
Lin said, “When you take out Npas4, the synaptic strength is almost saturated.”
“And then when learning takes place, although the memory-encoding cells can be fluorescently labeled, you no longer see the strengthening of those connections.”
In future work, Lin hopes to study how the circuit connecting the dentate gyrus to CA3 interacts with other pathways required for reminiscence retrieval.
She added, “Somehow there’s some crosstalk between different pathways so that once the information is stored, it can be retrieved by the other inputs.”