Neurodegenerative diseases, characterized by a progressive loss of brain function, result primarily from synaptic loss and neuronal cell death in the central nervous system. Alzheimer’s disease (AD) is arguably the most common neurodegenerative disorder leading to dementia. The etiology and pathogenesis of AD are incompletely understood, and effective, disease-modifying drug treatments are lacking.
Prior work has shown that genetic, environmental, and age-related factors, along with alterations in energy metabolism, autophagy, and synaptic function, all contribute to the pathogenesis of AD. A new study by scientists at Scripps Research and Massachusetts Institute of Technology (MIT) has found a clue to the molecular cause of Alzheimer’s. This clue may also explain why women are at greater risk for the disease.
Scientists discovered that the brains of women who had died from the condition had considerably greater concentrations of complement C3, a hazardous inflammatory response protein that has undergone chemical modification. Additionally, they demonstrated that estrogen typically protects against forming this type of complement C3.
Study senior author Stuart Lipton, MD, Ph.D., professor and Step Family Foundation Endowed Chair in the Department of Molecular Medicine at Scripps Research said, “Our new findings suggest that chemical modification of a component of the complement system helps drive Alzheimer’s, and may explain, at least in part, why the disease predominantly affects women.”
Lipton’s laboratory investigates biochemical and molecular processes, such as protein S-nitrosylation, which result in a modified version of complement C3, which may be the cause of neurodegenerative disorders. This chemical process, which produces a modified “SNO-protein” when a nitric oxide (NO)-related molecule attaches securely to a sulphur atom (S) on a specific amino acid building block of proteins, was first found by Lipton and his coworkers. Small clusters of atoms, like NO, frequently modify proteins in cells.
These modifications typically activate or inactivate a target protein’s functions. Because of technical difficulties, S-nitrosylation has received less attention than other protein modifications. Still, Lipton hypothesises that the “SNO-storms” of these proteins may play a significant role in the development of Alzheimer’s disease and other neurodegenerative diseases.
In the latest study, the scientists measured the number of proteins changed in 40 postmortem human brains using brand-new techniques for identifying S-nitrosylation. The brains were divided equally between males and females, with half coming from persons who had died of Alzheimer’s disease and the other half coming from people who hadn’t.
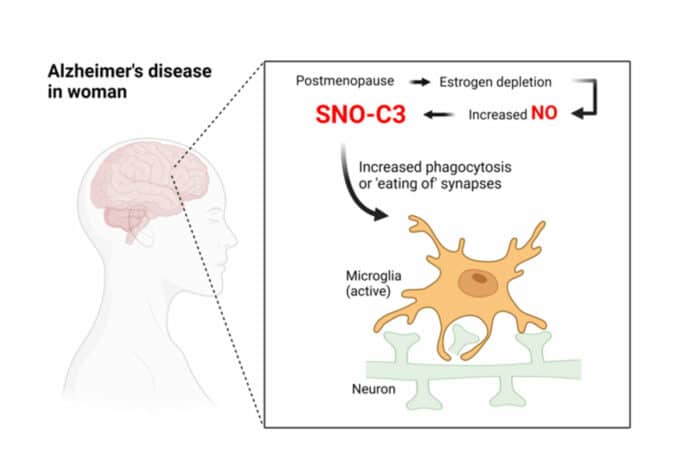
The scientists found 1,449 distinct proteins that had been S-nitrosylated in these brains. Numerous proteins that have already been connected to Alzheimer’s disease were among those that were most often altered, including complement C3. Surprisingly, female Alzheimer’s brains had S-nitrosylated C3 (SNO-C3) levels that were more than six times higher than those of male Alzheimer’s brains.
The complement system is an older component of the human immune system in terms of evolution. The “complement cascade” comprises a family of proteins, including C3, that can activate one another to cause inflammation. For more than 30 years, scientists have known that, when compared to neurologically healthy brains, Alzheimer’s brains exhibit higher amounts of complement proteins and other inflammatory indicators.
More recent studies have demonstrated, in particular, how complement proteins can cause brain-resident immune cells known as microglia to degrade synapses. At these junctions, neurons communicate with one another. Loss of synapses is a substantial correlate of cognitive decline in Alzheimer’s disease, and many scientists now assume that this synapse-destroying mechanism at least partially drives the disease process.
Why would female brains with Alzheimer’s have a higher prevalence of SNO-C3? The scientists proposed the hypothesis that estrogen explicitly protects women’s brains from C3 S-nitrosylation—and that this protection is lost when oestrogen levels sharply decline with menopause. There has long been evidence that the female hormone estrogen can have brain-protective effects under some circumstances.
This theory was validated by experiments using cultured human brain cells, which showed that SNO-C3 increases as estrogen (-estradiol) levels fall due to activating an enzyme that produces NO in brain cells. This rise in SNO-C3 triggers the degradation of synapses by microglia.
Study senior author Stuart Lipton, MD, Ph.D., said, “Why women are more likely to get Alzheimer’s has long been a mystery, but I think our results represent an important piece of the puzzle that mechanistically explains the increased vulnerability of women as they age.”
“Mechanistic insight into female predominance in Alzheimer’s disease based on aberrant protein S-nitrosylation of C3.”
Journal Reference:
- Hongmei Yang et al. Mechanistic insight into female predominance in Alzheimer’s disease based on aberrant protein S-nitrosylation of C3. Science Advances. DOI: 10.1126/sciadv.ade0764