Scientists at the Tokyo Institute of Technology, Japan, have demonstrated that reactions of alpha-hydroxy acids, like the alpha-amino acids that make up modern proteins, shape expansive polymers effectively under conditions assumed common on early Earth. These alpha-hydroxy acid polymers may have supported the development of living frameworks on early Earth.
This multi-national team demonstrated that hydroxy acids polymerize more effortlessly than amino acids and that they could have given the essential toolbox to kick-begin the development of more mind-boggling particles for the beginning of life on Earth.
There are diverse hypotheses for how life initially shaped on early Earth. One famous one proposes that life may have emerged in particular situations, for example, tidal pools or shallow water hot springs, where simple chemical reactions would have produced life’s forerunners. All life is comprised of polymers, huge atoms made up of an arrangement of particles called monomers. A key question is a way natural polymers could have shaped without compounds on early Earth.
While conditions of early Earth may have had monomers that could offer ascent to life, it would have been troublesome for polymers to emerge from these without the assistance of enzymes. For this situation, the group demonstrated these polymers could have framed with alpha-hydroxy acids before the presence of chemicals on early Earth.
Now, scientists in a new study showed that hydroxy acids polymerize more easily than amino acids. They could have given the essential toolbox to kick-begin the arrangement of more unpredictable particles for the inception of life on Earth.
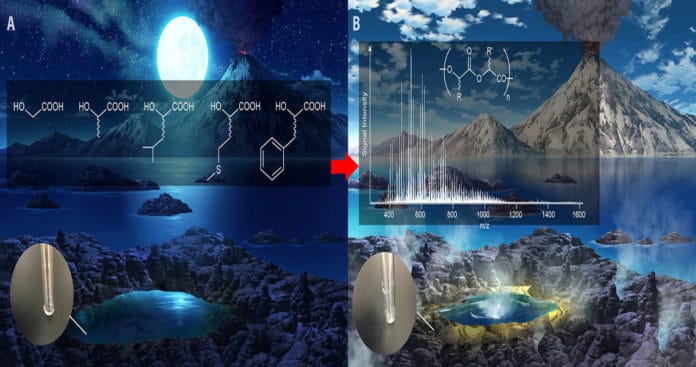
To mimic different primitive situations, the group reacted alpha hydroxy acids under fluctuated states of pH and temperature, from room temperature to boiling hot. Utilizing modern high-resolution mass spectrometry and data analysis software, they demonstrated that these polymers can shape over a scope of conditions.
The group indicated further, that utilizing a blend of various sorts of alpha-hydroxy acid that they could shape colossal quantities of kinds of polymers, basically making immense ‘libraries’ of various chemical structures, which can stay stable over a scope of natural conditions. The work finished with the production of a polyester library made up of five sorts of alpha hydroxy acids (Figure 1). They demonstrated that this easy-to-make library could contain several trillions of particular polymer groupings.
This examination opens new territories of investigation of the science that may have prompted the development of life. The creators are keen to broaden this work by exploring the kinds of elements of alpha-hydroxy corrosive polymers may have the capacity to do, and different sorts of simple synthetic substances that could interface with these, perhaps prompting self-repeating chemical frameworks.
The study is published in the journal Communications Chemistry.