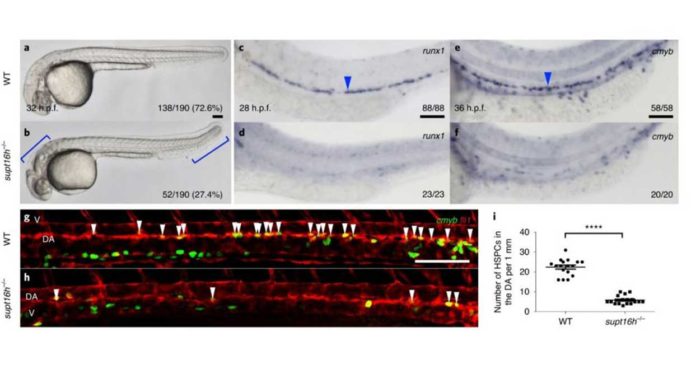
In collaboration with the University of California, San Diego (UCSD), Director Kyungjae Myung, and Dr. Yoonsung Lee at the Center for Genomic Integrity within the Institute for Basic Science (IBS) at UNIST, scientists have revealed the mechanism of development of hematopoietic stem cells (HSCs) that give rise to all blood cell types. Scientists demonstrated that Supt16h is a component of the Facilitates chromatin transcription (FACT) complex that is essential for forming hematopoietic stem and progenitor cells (HSPCs).
Hematopoietic foundational microorganisms (HSCs) are multipotent progenitors of blood cells that have self-renewal and proliferation abilities. Thus, severe control of the temporal and spatial specification maturation and extension of HSCs is crucial for the normal function of the blood and immune system. Notably, HSCs arise from the hemogenic endothelium, a special populace of endothelial cells inside the dorsal aorta’s ventral mass that transdifferentiates into HSCs.
In this study, scientists mainly focused on histone chaperone proteins to find a new gene responsible for HSPC formation. Histone chaperones are proteins that go about as molecular escorts to counter histone-DNA aggregation during nucleosome assembly. They also work as spools for the thread-like DNA to fold over to keep it tangled. When these proteins are expressed in the wrong place or at the wrong time, it modifies cell function and contributes to the movement of cancer and genetic diseases.
To understand how HSCs are specified, scientists performed a forward genetic screen using zebrafish embryos to discover novel genes needed for HSC specification, acquiring various mutants that neglected to shape HSCs in the aorta.
Scientists noted, “We are currently characterizing the mutant phenotypes and are mapping and positionally cloning genes, which cause the defects of HSC formation. Our studies will provide insight into the molecular cues that specify HSCs.”
Scientists mutated DNA in 13,000 zebrafish. They found that Supt16h plays a crucial role in developing hematopoietic stem cells via next-generation sequencing analysis. They also observed that zebrafish supt16h mutants express reduced levels of notch-signaling components, inhibiting HSC production.
Dr. Lee Yoon-sung at IBS said, “The study results have shown that the histone chaperone Supt16h regulates the expression of genes essential for the development of specific cells and that the notch-signaling system plays a key role in HSC production. We expect our findings to be a significant indicator for improving the treatment of blood diseases, including leukemia and blood cancer, by using HSC.”
Journal Reference:
- Sophia G. Espanola, Hyemin Song, Eunjin Ryu, et al., “Haematopoietic Stem cell-dependent Notch Transcription is Mediated by p53through the Histone Chaperone Supt16h,” Nature Cell Biology, (2020). DOI: 10.1038/s41556-020-00604-7