Peptides i.e., short amino acid chains monitor the main mechanism of the body. If using it for medications, it represents a billion-dollar market in the pharmaceutical industry. But, these peptides should be injected properly.
Now, scientists at the Technical University of Munich determined how peptides can be designed so that they can be easily administered as a liquid or tablet.
Horst Kessler, Carl von Linde Professor at the Institute for Advanced Study at TU Munich said, “Peptides are wonderfully well-suited as medication. The body already uses them as signaling molecules, and when they have done their job, they can be recycled by the body – no accumulation, no complicated detoxification.”
There are almost 500 peptide-based medications available for clinical trials. A modest bunch of peptide medicines is as of now charging incomes in the billions. In any case, the way that they can’t be regulated as tablets is a conclusive inconvenience of all substances in this classification.
Since proteins are an imperative piece of the eating regimen, the stomach and digestive organs harbor incalculable compounds that break peptide bonds. No drug in light of unmodified peptides would have an opportunity to survive the section through the gastrointestinal tract.

However, notwithstanding when fittingly adjusted peptide mixes endure the stomach in place, another obstacle anticipates them: The cells of the intestinal dividers keep their ingestion into the blood. That is the reason these sorts of dynamic operators are for the most part just be directed by infusion.
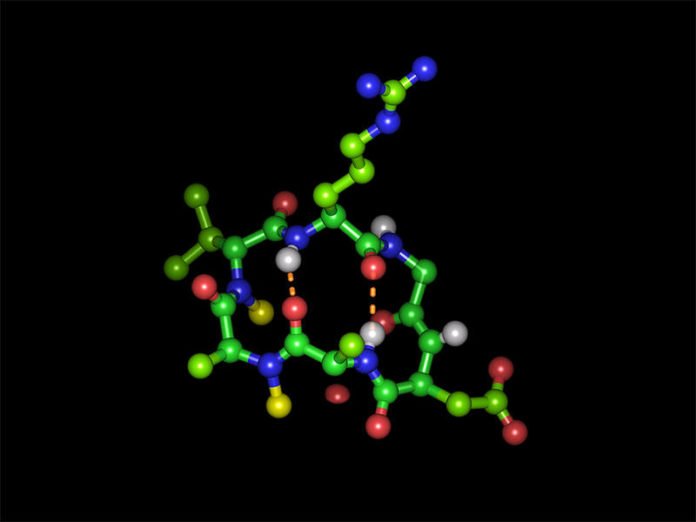
Scientists at very first approached this challenge by using round shaped peptide to investigate what effect replacing hydrogen atoms of the peptide bonds with methyl groups has on oral availability. It comprised six molecules of the simplest amino acid, alanine.
Prof. Kessler said, “It appears that cyclic hexapeptides with a specific structure are able to use an existing transport system.”
The group picked integrin receptors that control an assortment of capacities on the cell surface as an objective for their peptides. An arrangement of the three amino acids arginine, glycine and aspartic corrosive is the way to the docking at these receptors. Kessler’s collaborators consolidated the key grouping at various places of their model peptide, hence making new variations.
In any case, both the adversely charged side chain of aspartic corrosive and the decidedly charged arginine ended up being thump out criteria for utilizing the vehicle framework. The group, in any case, figured out how to veil the charged gatherings of both amino acids with securing gatherings.
Despite the fact that with this the peptide at first loses its capacity to tie to the objective particle, if the privilege defensive gatherings are chosen, they are divided from again by catalysts that are universal in the blood. The pharmaceutical impact is in this way reestablished upon landing in their goal.
Cell tests have shown that the new hexapeptide indeed has a biological effect. In low doses, it stimulates the growth of blood vessels. When mice are fed the masked hexapeptide, the effect is the same as in those that were injected with the unmasked hexapeptide.
Kessler said, “In the past, experts have designated the oral availability of peptide-based medications as the ‘holy grail of peptide chemistry.’ Our work provides a strategy for solving the challenges of stability, absorption in the body and biological effectiveness. In the future, this will greatly simplify the creation of peptide medication that can be easily given in fluid or tablet form.”
The compounds were designed, synthesized and tested for biological activity at the Technical University of Munich in Garching, structurally characterized at the CSIR National Chemical Laboratory in Pune (India) and at the Università di Napoli Federico II in Italy. Permeability was tested on cell systems at the Hebrew University in Jerusalem (Israel) and the biological effect in mice at Queen Mary University of London (UK).
The correspondence between the cells of a body in the different organs is a basic essential for the presence of multicellular living beings. Receptors on the phone surface, the supposed integrins, assume a critical part here. They pass on data about a cell’s domain to the cell inside.
Tumors, for instance, send development signs to vein cells to keep them developing and along these lines secure the blood supply to the tumor. What’s more, breaking down integrins are the reason for some illnesses making them exceedingly fascinating to pharmaceutical analysts.
Of the 24 human integrins, eight subtypes perceive a little grouping involving just three amino acids: arginine, glycine, and aspartic corrosive (shorthand: Arg-Gly-Asp, acronym: RGD).
This structure fits into the integrin receptor like a key. Flag atoms and proteins that have this arrangement trigger a response of the cell. Together with other amino acids that serve an acknowledgment part, the spatial structure of the arrangement figures out which of the integrins the key fits too.
Pharmaceutical analysts are currently watchful for atoms that have these acknowledgment successions in the fitting spatial structure, enabling them to trigger a similar cell response. The capacity to explicitly target diverse integrin subtypes is an imperative advance toward customized prescription in which a wide range of growth can be battled on a patient-particular premise.