Researchers in Japan and India have built up a reusable, superior catalyst in view of level formed ruthenium nanoparticles (Ru-NP) for the generation of profitable synthetic substances. Because of its exhibited strength, the catalyst could be generally utilized as a part of the vast scale generation of numerous kinds of colors, cleansers, agrochemicals, and pharmaceuticals.
Scientists have shown that nanoscale changes in surface structure can endlessly enhance the execution of metal catalyst utilized as a part of the generation of essential amines1, a vital class of mixes in the concoction business.
Contrasted with traditional metal-bolstered catalysts, the new catalyst, with its exceptionally dynamic surface structure, accomplished an overwhelmingly high reactant turnover.
The examination included looking at how well various types of metal catalysts could change over biomass-inferred furfural to furfurylamine. This filled in as a model response for reductive amination2, the fundamental procedure used to yield essential amines.
Michikazu Hara and Keigo Kamata at Tokyo Tech‘s Institute of Innovative Research said, “To the best of our knowledge, the Ru-NP catalyst exhibited the highest turnover frequency among the metal catalysts reported to date for the reductive amination of furfural to produce furfurylamine.”
Scientists noted, “Further experiments to test the reusability of the new catalyst for the reductive amination of furfural showed “no noticeable decrease in activity, even after four reuses. It thus highlights the catalyst’s robustness.”
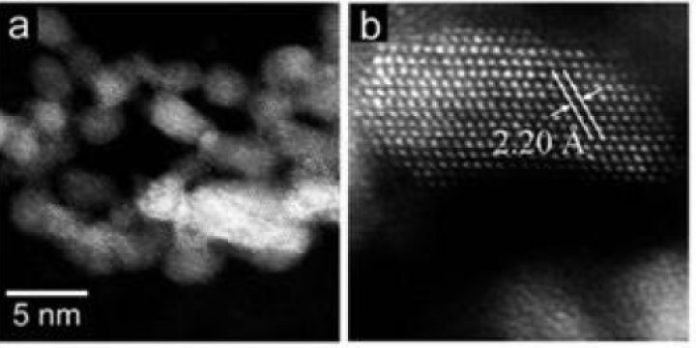
The mystery behind the catalysts’ uncommon execution lies in its particular structure and shape. More often than not, Ru has a hexagonal close-packed structure3 that points of catalytic performance. Conversely, the new catalyst has a high extent of supposed level molded, pristine face-centered-cubic4 ruthenium nanoparticles at the surface, which serve as active sites with weak electron-donating ability. It is this element that is believed to be basic to empowering proficient and specific reductive amination of different carbonyl mixes.
And also encouraging the creation of biomass-inferred amines, the new impetus could likewise help meet the requests of the more extensive fine substance industry.
The study is published in the journal Chemical Science.