Researchers from the National University of Singapore (NUS) have created a whole new library of atomically thin, two-dimensional (2D) materials using a novel and powerful approach of engineering the composition of transition metal dichalcogenides.
Materials that are atomically thin offer a platform to explore a wide range of intriguing physical properties and could provide many future applications. For example, ‘transition metal dichalcogenide monolayers’ are atomically thin semiconductors which are tipped to bring about the next generation of transistors, solar cells, LEDs, and more.
Transition metal dichalcogenide monolayers take the form MX2, with ‘M’ being a metal atom from the transition block of the periodic table and ‘X’ being a chalcogen atom (such as S, Se, or Te). However, fine-tuning the composition of 2D transition metal dichalcogenides to make new materials other than the standard compounds is usually challenging.
Now, a research team led by Professor Loh Kian Ping from the NUS Department of Chemistry, and Professor Stephen J. Pennycook from the NUS Department of Materials Science and Engineering, has synthesised and characterised for the first time, an atlas of atomically thin materials based on inserting the same metal atom between two transition metal dichalcogenides monolayers.
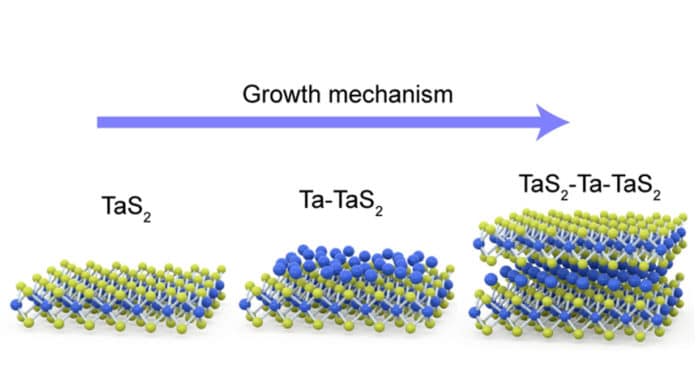
This insertion is known as intercalation, hence the researchers have named this new library ‘ic-2D’, to denote a class of materials where the atoms ‘intercalate’ themselves into the gap between the layers of crystals.
The researchers’ results were published in Nature on 13 May 2020.
Creation of a new atomically thin material
“If we splice two layers of transition metal dichalcogenide a little apart, we can see the chalcogen sites have slots like an egg holder. Another layer of metal atoms can occupy the slots in the same way we can arrange eggs in the egg holder. This is the magic of ic-2D materials,” explained Prof Pennycook.
This is a new way of thinking when it comes to transition metal dichalcogenides. In the past, theoreticians tried predicting new properties based the traditional bonding sites of metal and chalcogen atoms in the material. However, their theories did not address the situation when the same metal atom sits in the gap between two crystals.
As such, the research team developed a way to synthesis the novel materials by providing conditions where the metal atoms are in excess of the chalcogens. In this way, more than 10 different types of ic-2D materials have been experimentally discovered by the team, some of which are ferromagnetic.
Discovering the library of ic-2D materials
Theoretical calculations performed by the team have shown that the ‘self-intercalation’ method the team developed is applicable to a large class of 2D layered materials. This means that there is a new library of ic-2D materials waiting to be discovered.
“This new method for engineering the composition of a broad class of transition metal dichalcogenides offers a powerful approach to transform layered 2D materials into ultra-thin, covalently bonded ic-2D crystals with ferromagnetic properties. This technique is expected to be compatible with most material growth methods,” said Prof Loh, who is also from the NUS Centre for Advanced 2D Materials.
Dr Zhao Xiaoxu, the first author of the paper, unveiled the novel materials with an electron microscope, and found that the intercalated metal atoms consistently occupy the same vacancies resulting in distinct patterns depending on the intercalation concentrations.
Prof Loh commented, “With versatility in composition control, we have shown that it is possible to tune, in one class of materials, properties that can vary dramatically. This discovery presents a rich landscape of ultra-thin 2D materials that await the further discovery of new properties.”
Next steps
Moving forward, the research team plans to incorporate this new library of materials into memory devices for practical applications, and intercalate foreign atoms to exploit novel functionalised ic-2D materials.
Journal
- Xiaoxu Zhao, Engineering covalently bonded 2D layered materials by self-intercalation. DOI: 10.1038/s41586-020-2241-9