The “CaV2.2” channel, one of the voltage-dependent calcium channels that control the inflow of calcium ions and is expressed at the axon terminal of nerve cells to control neurotransmitter production, is crucial for signal transmission between nerve cells. Mental disorders like bipolar disorder, schizophrenia, autism, epilepsy, and chronic pain have been linked to issues with CaV2.2 activity regulation.
Phospholipids are the building blocks of the cell membrane. It is well established that the phospholipid PIP2 is crucial for the function of voltage-dependent calcium channels. However, the exact mechanism by which PIP2 controls calcium channel function has not been determined because of the intricate structure in which several subunits are bound.
Professor Suh Byung-chang’s research team at the Department of Brain Sciences, DGIST, identified the regulation mechanism of calcium channels, which are essential for signal transmission between nerve cells. They studied the molecular mechanism of PIP2 concerning the activation of various receptors and ion channels.
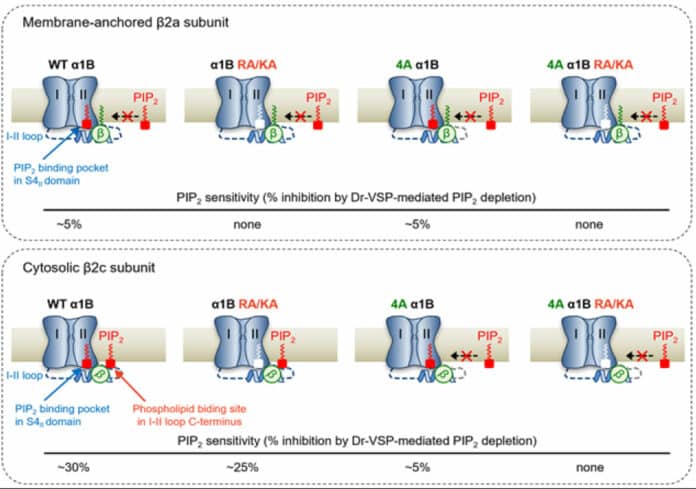
Scientists, in particular, have demonstrated that the voltage-dependent calcium channel’s sensitivity to PIP2 depends on the degree to which its auxiliary subunit, known as the 2 unit, is attached to the cell membrane.
These investigations served as the foundation for this study, which sought to determine the fundamental mechanism by which PIP2 regulates CaV2.2 activity differently depending on whether the 2 unit is physically or molecularly linked to the cell membrane. The research team used genetic recombination to develop several mutant models of CaV2.2 channels and two units, which were then verified by electrophysiological methods.
As a result, it was found that PIP2 binds to the ‘I-II loop,’ to which β2 units bind, in the CaV2.2 channel and to ‘S4II’, one of the voltage sensing domains, respectively, to regulate the activity of the channel. In addition, it was found that the binding of PIP2 to the I-II loop is determined depending on whether the β2 unit binds to the cell membrane, thereby regulating the activity of CaV2.2.
Furthermore, it was confirmed that CaV2.2 activity could be regulated in real-time by developing a system that can artificially control the binding of PIP2 to the I-II loop of CaV2.2 by applying β2 unit.
Scientists noted, “The result identified a new action mechanism for CaV2.2 channel activity, which plays a vital role in signal transmission between nerve cells. It is expected to provide an important clue to the treatment of mental disorders, such as autism, bipolar disorder, and schizophrenia, and of fatal neurological diseases, such as epilepsy and chronic pain in the future.”
Journal Reference:
- Cheon-Gyu Park, Wookyung Yu, Byung-Chang Suh, Molecular basis of the PIP2-dependent regulation of CaV2.2 channel and its modulation by CaV β subunits. eLife. DOI: 10.7554/eLife.69500