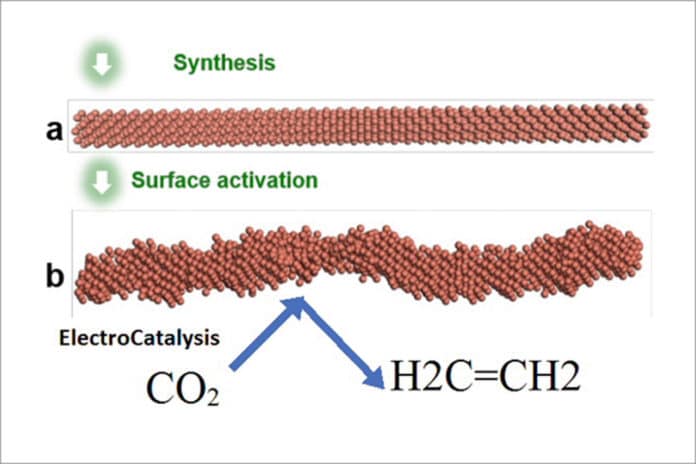
Scientists from Caltech and the UCLA Samueli School of Engineering have come up with a promising way to convert carbon dioxide into ethylene.
By developing nanoscale copper wires with specially shaped surfaces, scientists catalyzed a chemical reaction that reduces greenhouse gas emissions while generating ethylene—a valuable chemical simultaneously.
The results demonstrated that he shaped catalyst favors the production of ethylene over hydrogen or methane.
Yu Huang, the study’s co-corresponding author, said, “We are at the brink of fossil fuel exhaustion, coupled with global climate change challenges. Developing materials that can efficiently turn greenhouse gases into value-added fuels and chemical feedstocks is a critical step to mitigate global warming while turning away from extracting increasingly limited fossil fuels. This integrated experiment and theoretical analysis present a sustainable path towards carbon dioxide upcycling and utilization.”
William A. Goddard III, the study’s co-corresponding author and Caltech’s Charles and Mary Ferkel Professor of Chemistry, Materials Science, and Applied Physics, said, “The idea of using copper to catalyze this reaction has been around for a long time, but the key is to accelerate the rate, so it is fast enough for industrial production. This study shows a solid path towards that mark, with the potential to transform ethylene production into a greener industry using CO2 that would otherwise end up in the atmosphere.”
Using copper to kick start the carbon dioxide (CO2) reduction into ethylene reaction (C2H4) has suffered two strikes against it. First, the initial chemical reaction also produced hydrogen and methane—both undesirable in industrial production. Second, previous attempts that resulted in ethylene production did not last long, with conversion efficiency tailing off as the system continued to run.
Scientists overcome these hurdles by designing copper nanowires with highly active “steps”—similar to a set of stairs arranged at the atomic scale.
One intriguing finding of this collaborative study is that this step pattern across the nanowires’ surfaces remained stable under the reaction conditions, contrary to the general belief that these high energy features would smooth out. This is the key to both the system’s durability and selectivity in producing ethylene, instead of other end products.
The new system ran for 200 hours, with little conversion efficiency, a significant advantage for copper-based catalysts. The comprehensive understanding of the structure-function relation illustrated a new perspective to design highly active and durable CO2 reduction catalysts in action.
Journal Reference:
- Chungseok Choi et al., Highly active and stable stepped Cu surface for enhanced electrochemical CO2 reduction to C2H4, Nature Catalysis (2020). DOI: 10.1038/s41929-020-00504-x