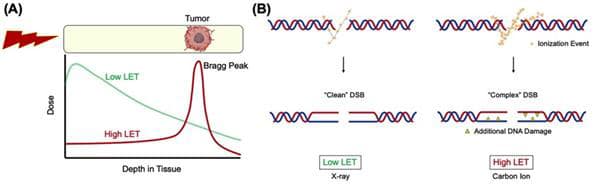
Cancer patients typically get ionizing radiation therapy (IR), which is thought to work by causing DNA breaks in the cells. Particle accelerators emit radiation from heavily charged particles like carbon ions, which have a high linear energy transfer (LET) and release most of its energy over a small area known as the Bragg peak. Compared to routinely utilized low LET radiation like gamma or x-rays, damage to nearby normal tissues can be reduced if the Bragg peak is focused on the tumor.
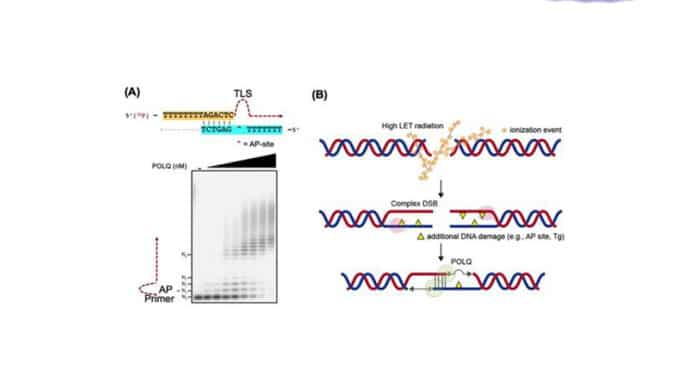
High LET heavy ion radiation (hiLET) generates DNA double-strand breaks (DSBs) that are more “complex” than low LET-induced DSBs (loLET-DSBs). They cause additional DNA damage, such as apurinic/apyrimidinic (AP) site and thymine glycol (Tg) near the DSB sites. This makes it more cytotoxic per unit dose than low LET radiation.
However, it remains to be seen how these hiLET-DSBs are processed in mammalian cells.
UNIST scientists visited the Japan QST hospital to use the HIMAC (Heavy Ion Medical Accelerator in Chiba) synchrotron for high LET radiation. They theorized that DNA polymerase θ (POLQ) is essential in repairing complex DSBs.
POLQ is a special DNA polymerase that can carry out translesion synthesis (TLS) through an abasic (AP) site and thymine glycol (Tg) as well as microhomology-mediated end-joining. The biological significance of the TLS activity was unknown, though.
Ms. Yubin Sung, one of the joint first authors, said, “We provided evidence that the TLS activity of POLQ plays a critical role in repairing hiLET-DSBs. We found that POLQ efficiently anneals and extends substrates mimicking complex DSBs.”
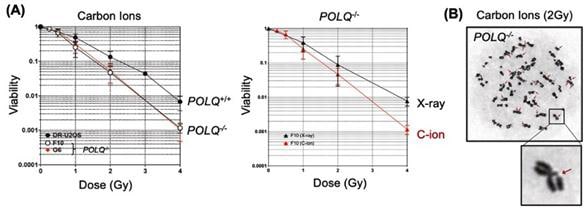
Mr. Geunil Yi, another joint first author, said, “We demonstrated that genetic disruption of POLQ results in an increase of chromatid breaks and enhanced cellular sensitivity following treatment with high LET radiation.”
“At the biochemical level, POLQ could bypass an AP site and Tg during end-joining and annealing two single-stranded DNA tails when DNA lesions were located outside the microhomology.”
The single-molecule FRET assay technique was created with Prof. Hajin Kim and Mr. Chanwoo Kim at UNIST to track POLQ-mediated annealing and DNA extension. At IBS-CGI, Ms. Jae Sun Ra examined chromatid breakage from high LET radiation. The tests with HIMAC were carried out with Prof. Fujimori and Mr. Hirakawa at QST, as well as Prof. Kato at Colorado State University.
Professor Kei-ichi Takata, Center for Genomic Integrity, Institute for Basic Science, said, “We are proud to announce the publication of our paper, which was only possible through the great teamwork of everybody involved. Our findings provide new insights into the mechanisms of how hiLET-DSB is repaired in mammalian cells and further suggest that the inhibition of POLQ may augment the efficacy of heavy ion radiation therapy.”
Journal Reference:
- Geunil Yi, Yubin Sung, Chanwoo Kim, et al. “DNA polymerase θ-mediated repair of high LET radiation-induced complex DNA double-strand breaks,” Nucleic Acids Research (2023). DOI: 10.1093/nar/gkad076