A healthy immune system strikes a harmony between destroying infections and cancers and not blowing up to harm one’s own particular tissue. Immune checkpoints help control the resistant reaction, yet tumors misuse these checkpoint pathways by communicating exceptional proteins that sidestep antitumor insusceptible reactions. One noteworthy checkpoint inhibitor pathway is the PD-1 pathway, and its ligand is PD-L1.
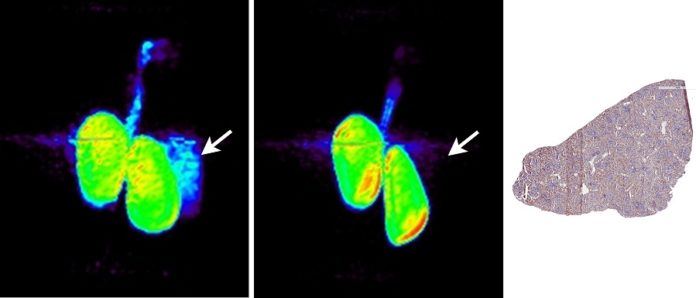
Now SNMMI scientists reported that they have devised same-day, noninvasive positron emission tomography (PET)-based imaging approach to assess PD-L1 positive tumors. In this approach, the PD-L1 ligand, which enables cancer to evade a person’s immune system, has been successfully targeted for the first time with a fluorine-18 (18F)-labeled PD-L1 radioligand.
David J. Donnelly, Ph.D., at Bristol-Myers Squibb Research said, “This approach represents an opportunity for physicians to noninvasively assess all of a patient’s tumors for PD-L1 expression with a single PET scan and timely readout. This may help guide treatment decisions and assess treatment response, to help identify the right treatment for the right patient at the right time and right dose.”
Scientists labeled anti-PD-L1 adnectin with 18F to generate 18F-BMS-986192. They then evaluated it for distribution, binding, and radiation dosimetry in healthy cynomolgus monkey. Scientists found that the feasibility of the approach and the radiation dosimetry estimates indicate that the tracer is safe to administer in human studies.
The study is presented in the featured article of The Journal of Nuclear Medicine’s March issue.