One of the key highlights of breast cancer is that most patients endure if the disease stays local, however there is a more than 70% drop in survival if the cells have metastasized. Notwithstanding, when the cells leave the primary tumor, they are frequently not responsive to the drugs that initially worked for the patient.
Scientists at Purdue University wanted to develop a system that could help them better understand how the physiology of a new tissue space affected tumor cells upon invasion into the new organ.
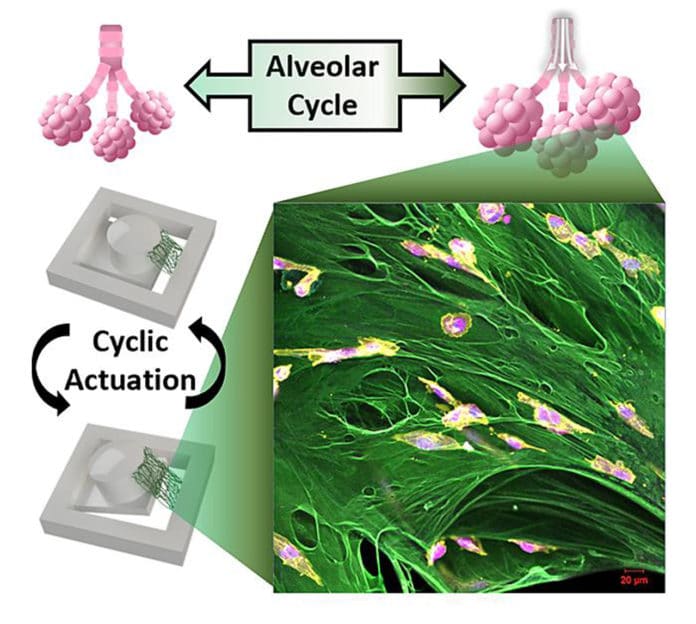
Scientists have developed a novel testing platform to evaluate how breast cancer cells respond to the recurrent stretching that occurs in the lungs during breathing. The technology is designed to better understand the effects that the local tissue has on metastatic breast cancer to study how metastases grow in new tissue.
In this magnetically moving cell culturing system, the cancer cells can be grown in 3D on a suspended extracellular matrix protein that is abundant in early metastatic lung tissue in order to evaluate the impact of mechanical forces.
They were able to incorporate the strain amplitude and rate of breathing in this tissue mimic. The scientists found that the cells quit dividing under these conditions.
Michael Wendt, a Purdue associate professor of medicinal chemistry and molecular pharmacology said, “Never before has the concept of motion been interrogated as a component of the tumor microenvironment. We now understand that healthy organs utilize motion to resist metastatic colonization. The development of this microactuator system will not only continue to yield increased biological understanding, of metastasis, but it will also serve as a platform for us to better evaluate pharmacological inhibitors of the most lethal aspect of cancer progression.”
Hyowon “Hugh” Lee, an associate professor of engineering and a researcher at the Birck Nanotechnology Center said, “This is the first attempt to engineer a cell culture system that can apply mechanical forces on a suspended tissue. Most bioreactors with mechanical stimulation capabilities rely on growing 2D cell culture on flat non-biological substrates, but we are using a custom magnetic actuator and suspending a layer of fibronectin to grow 3D cancer cells like a miniature tissue.”
“Our system better mimics the physiological environment without using artificial substrates. Using this platform, we show that certain cancer cells slow down their proliferation due to the cyclic stretching of breathing.”
Sarah Calve, a Purdue adjunct professor of biomedical engineering, and Adrian Buganza Tepole, a Purdue assistant professor of mechanical engineering, interfaced with the mechanical characteristics of the stretching protein. They measured the response of the material to stretching and developing a mapping of the strains felt by the cancer cells at various locations on the device.
Angel Enriquez, a doctoral student in Lee’s lab, said, “One key takeaway has been the benefits of collaboration with people outside of your field of expertise and how they can provide more complete research.”
Sarah Libring, a doctoral student and a co-first author from Solorio’s Lab, said, “It’s been amazing to be part of the development of a new device like this because by bringing together the expertise of multiple professors and multiple labs, we are now able to study cancer cells on dynamically moving fibronectin fibrils that hasn’t been previously possible.”
Journal Reference:
- Ángel Enríquez et al. High‐Throughput Magnetic Actuation Platform for Evaluating the Effect of Mechanical Force on 3D Tumor Microenvironment. DOI: 10.1002/adfm.202005021