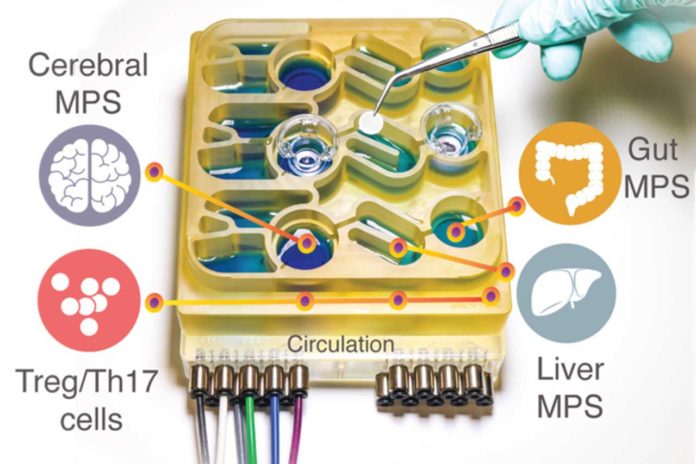
A new ‘organs-on-a-chip’ system, developed by MIT scientists- replicates interactions between the brain, liver, and colon. The system helps scientists model the impact that microbes living in the gut have on healthy brain tissue and tissue samples derived from Parkinson’s disease patients.
They found that short-chain fatty acids, produced by microbes in the gut and transported to the brain, can have different impacts on healthy and diseased brain cells.
Martin Trapecar, an MIT postdoc and the lead author of the study, said, “While short-chain fatty acids are largely beneficial to human health, we observed that under certain conditions they could further exacerbate certain brain pathologies, such as protein misfolding and neuronal death, related to Parkinson’s disease.”
In a past study, scientists used a microphysiological system to model interactions between the liver and the colon. In that study, they found that short-chain fatty acids (SCFAs), molecules produced by microbes in the gut, can worsen autoimmune inflammation associated with ulcerative colitis under certain conditions. In this study, scientists decided to add the brain and circulating immune cells to their multiorgan system.
A few years ago, a study found the link between SCFAs and Parkinson’s disease in mice. It was shown that SCFAs produced by bacteria as they consume undigested fiber in the gut sped up the disease’s progression, while mice raised in a germ-free environment were slower to develop the disease.
Scientists, in this study, decided to further explore that study by using their microphysiological model.
Almost 20% of Parkinson’s cases have a genetic cause. The cells that scientists used for their Parkinson’s model carry a mutation that causes the accumulation of a protein called alpha-synuclein, which damages neurons and causes inflammation in brain cells. The brain cells were also generated with this mutation corrected but genetically identical and from the same patient as the diseased cells.
When scientists studied these two sets of brain cells in microphysiological systems that were not connected to any other tissues, they found that Parkinson’s cells showed more inflammation than the healthy, corrected cells. The Parkinson’s cells also had impairments in their ability to metabolize lipids and cholesterol.
Scientists then connected the brain cells to tissue models of the colon and liver, using channels that allow immune cells and nutrients, including SCFAs, to flow between them. They found that being exposed to SCFAs is beneficial for healthy brain cells and helps them mature. However, when brain cells derived from Parkinson’s patients were exposed to SCFAs, the beneficial effects disappeared. Instead, the cells experienced higher levels of protein misfolding and cell death.
These effects were seen even when immune cells were removed from the system, prompting scientists to hypothesize that lipid metabolism changes mediate the impact.
Trapecar said, “It seems that short-chain fatty acids can be linked to neurodegenerative diseases by affecting lipid metabolism rather than directly affecting a certain immune cell population. Now the goal for us is to try to understand this.”
Scientists are now planning to model other types of neurological diseases that may be influenced by the gut microbiome.
Linda Griffith, the School of Engineering Professor of Teaching Innovation, said, “The findings offer support for the idea that human tissue models could yield information that animal models cannot.”
DARPA funded the research, the National Institutes of Health, the National Institute of Biomedical Imaging and Bioengineering, the National Institute of Environmental Health Sciences, and the Koch Institute Support (core) Grant National Cancer Institute, and the Army Research Office Institute for Collaborative Biotechnologies.
Journal Reference:
- Martin Trapecar et al. Human psychomimetic model integrates microphysiological systems of the gut, liver, and brain to study neurodegenerative diseases. DOI: 10.1126/sciadv.abd1707