Inflammation is required for protective responses against pathogens and is thus essential for survival, but sustained inflammation can lead to diseases such as atherosclerosis and cancer. There are several treatments, but their action is often not very targeted, high doses are required, and deleterious side effects are frequent.
A team from the University of Geneva (UNIGE) and the Ludwig Maximilians Universität München (LMU) has succeeded in developing a fully biodegradable nanoparticle that can make an anti-inflammatory drug much more effective and less toxic. The nanoparticle can deliver drug directly into macrophages, ensuring its effectiveness.
Thanks to an in vitro screening methodology, scientists in this study eliminated the need for animal testing. The study could potentially lead to a potent and targeted anti-inflammatory treatment.
The new molecule called Necrosulfonamide (NSA) inhibits the release of several important pro-inflammatory mediators. It hence acts as a promising advance to reduce certain types of inflammation. However, being extremely hydrophobic, it travels poorly in the bloodstream and could target many cell types, triggering potentially toxic effects.
Gaby Palmer, a professor in the Department of Medicine and the Geneva Centre for Inflammation Research at the UNIGE Faculty of Medicine, said, “This is why this molecule is not yet available as a drug. Using a nanoparticle as a transport vessel would circumvent these shortcomings by delivering the drug directly into macrophages to combat inflammatory overactivation in the place where it begins.”
The primary criteria used by the scientists while testing various porous nanoparticles include a decrease in toxicity and dosage requirements, as well as the capacity to release the medication only after the nanoparticle had entered the macrophages’ core.
Carole Bourquin, a professor at the UNIGE’s Faculties of Science, who codirected this work at UNIGE, said, “We used an in vitro screening technology which we developed a few years ago on human and mouse cells. This saves time and greatly reduces the need to use animal models. Thus, only the most promising particles will be tested on mice, which is a prerequisite for clinical trials on humans.”
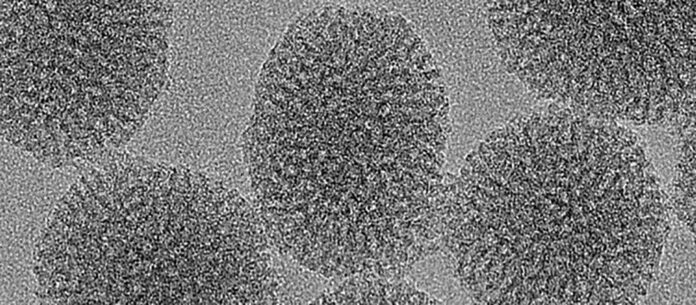
Bart Boersma, a doctoral student in Carole Bourquin’s laboratory and first author of this study, said, “Three very different nanoparticles featuring high porosity were examined: a cyclodextrin-based nanoparticle, a substance commonly used in cosmetics or industrial food, a porous magnesium phosphate nanoparticle, and finally a porous silica nanoparticle. The first was less satisfactory in cell uptake behavior, while the second proved counterproductive: it triggered the release of pro-inflammatory mediators, stimulating the inflammatory reaction instead of fighting it.”
“The porous silica nanoparticle, on the other hand, met all the criteria: it was fully biodegradable, of the right size to be swallowed by macrophages, and was able to absorb the drug into its numerous pores without releasing it too early. The anti-inflammatory effect was remarkable.”
Scientists then replicated their tests by coating the nanoparticles with an additional lipid layer, but with no greater benefit than silica nanoparticles alone.
Carole Bourquin said, “Other silica nanosponges developed by the German-Swiss team had already proven their effectiveness in transporting anti-tumor drugs. They carry a very different drug that inhibits the immune system.”
“Mesoporous silica is increasingly revealing itself as a nanoparticle of choice in the pharmaceutical field, as it is very effective, stable, and non-toxic. Nevertheless, each drug requires a tailor-made carrier: the shape, size, composition, and destination of the particles must be reassessed each time.”
Journal Reference:
- Bart Boersma, karin Moller, et al. Inhibition of IL-1β release from macrophages targeted with necrosulfonamide-loaded porous nanoparticles. Journal of Controlled Release. DOI: 10.1016/j.jconrel.2022.09.063