In recent years, the field of electron-based reactions has witnessed significant advancements, with researchers exploring ways to optimize and enhance the efficiency of these reactions. One promising approach that has gained attention is the utilization of slow electrons. By manipulating the energy and velocity of electrons, scientists have discovered that slowing them down can lead to more efficient and controlled reactions.
This exciting development opens up new possibilities for various applications, ranging from chemical synthesis to energy conversion and storage. In this article, we delve into the potential benefits of using slow electrons and examine how they can revolutionize the landscape of modern reaction engineering.
An international research team aimed to detect dielectrons, a peculiar chemical entity consisting of two electrons without a nucleus that had previously evaded direct detection. Led by Professor Ruth Signorelli from ETH Zurich, the team made an unexpected discovery during their investigation—a groundbreaking method to generate slow electrons that can trigger specific chemical reactions.
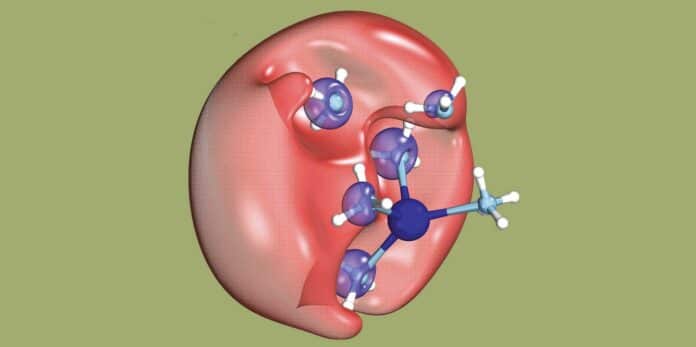
Dielectrons are inherently unstable, disintegrating back into separate electrons within a fraction of a nanosecond. However, the researchers demonstrated a fascinating phenomenon where one electron remains stationary while the other, possessing low energy and reduced speed, moves away. This innovative approach allows precise manipulation of the electron’s kinetic energy, effectively controlling its velocity.
Dielectrons, composed of two electrons without a nucleus, occupy small cavities. Scientists have discovered a method to create dielectrons by dissolving sodium in liquid ammonia and exposing the solution to ultraviolet (UV) light. When the UV light interacts with the ammonia and sodium, one electron from each combines to form a dielectron temporarily. Interestingly, when the dielectron separates, one of the electrons moves away at speed determined by the UV light’s wavelength, indicating energy transfer from the light to the electron.
Led by researchers from ETH Zurich and collaborating with scientists from the University of Freiburg, SOLEIL synchrotron, and Auburn University, this study holds significance in understanding the damage caused by low-energy electrons to human tissues, such as X-rays or radioactivity. These mobile electrons can interact with DNA and initiate chemical reactions.
Researchers can gain insights into radiation damage mechanisms by simplifying the production of slow electrons in a solution using UV light and controlling their energy levels. Additionally, this research has broader implications, as accepting a free electron is involved in various processes like the production of cortisone and other steroids. By facilitating the generation of slow electrons using UV light and controlling their energy, chemists can potentially enhance and optimize these reactions.
In conclusion, the discovery of slow electrons and their utilization in various reactions holds great promise for achieving enhanced efficiency and control. From radiation damage studies to synthetic processes, the ability to generate and manipulate slow electrons opens up new possibilities for scientific exploration and optimization of chemicals.
Journal Reference:
- Sebastian Hartweg, Jonathan Barnes, Bruce L. Yoder et al. Solvated dielectrons from optical excitation: An effective source of low-energy electrons. Science. DOI: 10.1126/science.adh0184