Water and other aqueous solutions in the sorts of volumes we deal with every day normally freeze when cooled below the freezing point of 0° C or 32° F.
Scientists at the Massachusetts General Hospital Center for Engineering in Medicine (MGH-CEM) have come up with a new technique that keeps liquid from freezing even at extremely low temperatures. The method called ‘Deep Supercooling’ works by blocking the interface between water and air.
Originally, the method is simply to cover the surface of such a liquid with a solution that does not mix with water, like mineral oil, thus avoiding the interaction of air and water. The method someday is expected to enable safe, extended preservation of blood cells, tissues, and organs, along with improved food preservation. Moreover, it can enable many medical and food preservation methods, as well as fundamental experiments that were not previously possible.
Reducing the temperature of any biological material – such as cold storage of perishable foods and organs for transplantation – slows down metabolic and other reactions. Supercooling extends this metabolic deceleration further without the damage caused by ice crystallization.
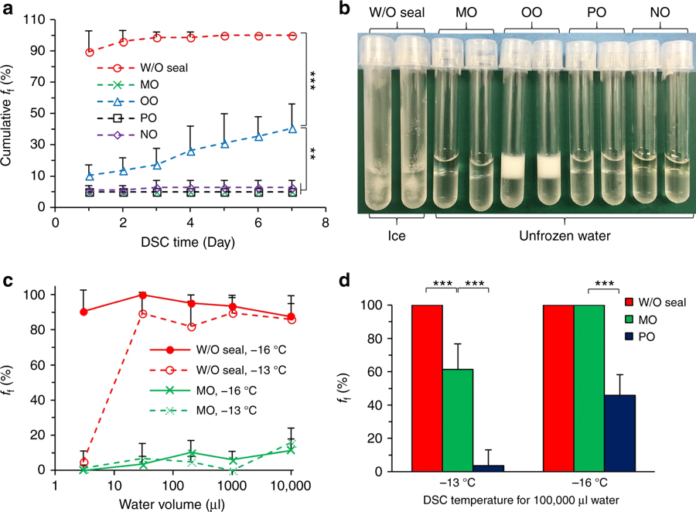
Observations showed that sealing the surface of a small (1 ml) water sample with a hydrocarbon-based oil – such as mineral oil, olive oil or paraffin oil – could suppress ice formation at temperatures as low as -13° C (around 9° F) for up to a week.
Through a progression of tests both with more mineral oils and with pure simple hydrocarbons, for example, alcohols and alkanes, they prevailing with regards to keeping 1 ml samples of water and cell suspensions supercooled at – 20° C (- 4° F) for 100 days and 100 ml (3.2 oz) tests for seven days.
The team also demonstrated the application of their deep supercooling method to the extended preservation of red blood cells. While red blood cells are usually stored at 4° C (39° F) for as long as 42 days.
O. Berk Usta, Ph.D., of the MGH-CEM, said, “We currently are conducting experiments to increase the volume of red blood cell storage samples up to the more clinically relevant 300 to 500 ml range. We also are working on applying this method to other cells and on translating it to large tissues and whole organs like the liver.”
“Along with potential applications in medicine and food preservation, we also believe this invention could be used to study chemical reactions in the liquid state at low temperatures without the usual costly and complicated high-pressure equipment.”
The study is published in the journal Nature Communications.