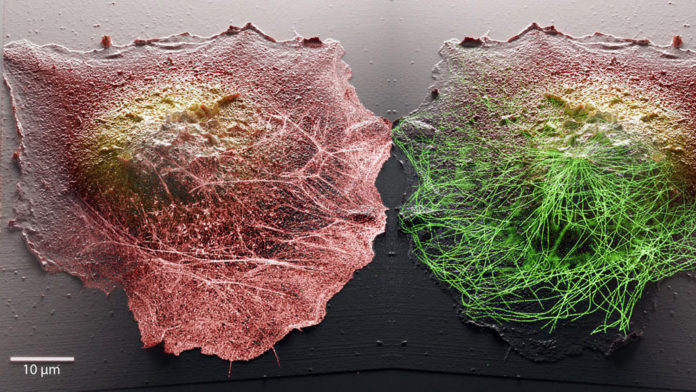
Understanding certain disorders and diseases and then developing effective treatments is essential to understanding cell functioning. But, observing living cells on a micro- or nanoscale remains a significant challenge.
By combining two different microscopy methods, EPFL scientists from two other labs have jointly developed a system to observe how living cells work with unparalleled precision. Scientists combined two complementary microscopies to develop their method to observe the cell surface and molecular activity inside.
Existing methods to observe live cells have many technical challenges.
Georg Fantner, the head of EPFL’s Laboratory for Bio- and Nano- Instrumentation (LBNI), said, “Techniques such as electron microscopy allows unmatched resolution of the cell surface at the nanoscale, but it requires placing samples under vacuum and bombarding them with electrons. Living organisms cannot survive that kind of treatment. Another common method is fluorescence microscopy. Although it lets you observe samples without destroying them, having sufficient resolution to resolve the three-dimensional surface of the cell is difficult. Additionally, the dose of photons required can cause cell damage.”
Scientists in this study coupled stochastic optical fluctuation imaging (SOFI) with scanning probe microscopy. Scanning probe microscopy generally involves touching a cell sample directly with a probe tip to reveal its surface and map out its topography.
Scanning probe microscopy generally involves touching a cell sample directly with a probe tip to reveal its surface and map out its topography. However, the mechanical contact between the sample and the tip is detrimental to the observation of live cells because it disturbs the cells’ native state.
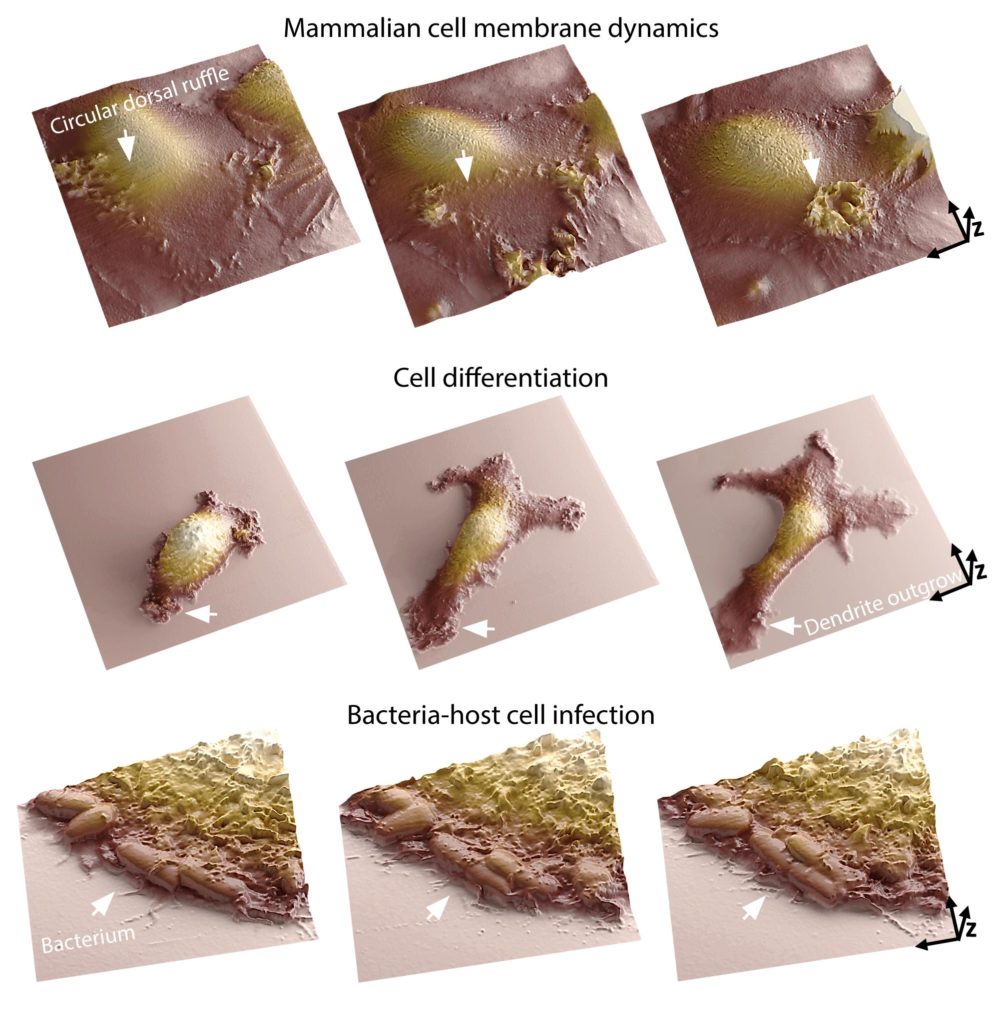
To develop their system, scientists replaced the physical probe with a glass nanopore that measures the flow of ions to detect the cell surface without contact.
Thanks to their system, scientists were able to see the interior and exterior of cells simultaneously. It also offers them a detailed insight into the links between phenomena occurring simultaneously in those two different places.
Samuel Mendes Leitão, a Ph.D. student at LBNI that developed the SICM microscope, said, “A cell’s membrane is the place where it interacts with its surroundings. It’s where many biological processes and morphological changes occur, like during cell infection. Our system lets researchers analyze molecular arrangements inside the cell and map out how they correlate with membrane dynamics. Moreover, we can now track those dynamics in great detail for temporal ranges of sub-second to days. Being able to image continuously at the nanoscale for long periods is one of the main challenges in live-cell microscopy, as cells are susceptible to small perturbations.”
Vytautas Navikas, a Ph.D. student at EPFL’s Laboratory of Nanoscale Biology (LBEN), developed the system’s optical components: “Another benefit of combining the two methods is that it incredibly improves image quality. We can now watch cellular processes with much greater resolution.”
Scientists noted, “The system can be used for observing phenomena like cell motility, differentiation and cell-cell communication, opens up many new avenues of research. It could be extremely useful in infection biology, immunology, and neurobiology – fields where it’s important to understand how a cell reacts in real-time to an outside stimulus.”
Journal Reference:
- Samuel M. Leitao, Barney Drake et al. “Time-Resolved Scanning Ion Conductance Microscopy for Three-Dimensional Tracking of Nanoscale Cell Surface Dynamics. DOI: 10.1021/acsnano.1c05202