An international team under the auspices of the University of Geneva (UNIGE) and Geneva University Hospitals (HUG) has tested a molecule inhibitor of interleukin-18, a protein involved in immune response. These promising upshots in terms of safety and efficacy are paving the way for a novel treatment, not only for Still’s disease but other rare inflammatory diseases, also.
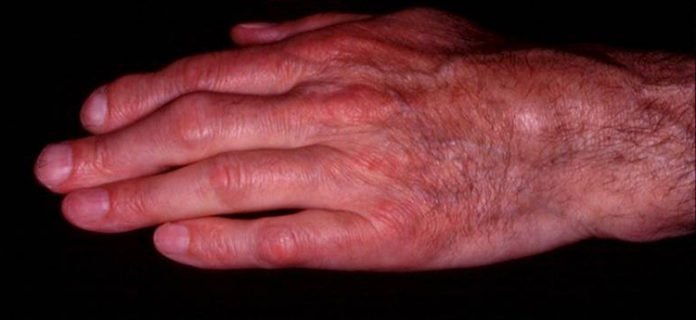
Still’s disease is a serious orphan disease manifested by high fevers, skin and joint involvement, including paralysis, as well as damage to other organs such as the liver or spleen. It is caused by a deregulation of the immune system triggering an acute inflammatory response.
Recently, a baby’s life was saved after the drug was administered as a last resort. This study published in the specialist journal Annals of the Rheumatic Diseases.
In its adult form, Still’s disease impacts about 1 in 100,000 people every year, with its infantile form being ten times more common. This infrequent condition can take different forms like monocyclic, polycyclic or chronic, leading to attacks on several organs that may threaten the quality of life, or even life, of those affected.
While the cause is still unknown, genetic factors have been identified in other, analogous syndromes.
Cem Gabay, a professor at UNIGE’s Faculty of Medicine and Head of the HUG Rheumatology Department, is one of the world’s leading specialists in these complex diseases and part of a European consortium whose aim is to comprehend the causes and pathology of these inflammatory conditions.
Cytokines are small proteins involved in cell-to-cell interactions, whose role in triggering certain diseases is now becoming clear. One of them, interleukin-18 (IL-18), specialises in immune and inflammatory responses.
A few years ago, a pharmaceutical company developed an injectable form of the IL-18 inhibitor, with the idea of offering treatment to combat rheumatoid arthritis and psoriasis. The tests proved inconclusive and the formula abandoned.
Andrew Sleight, CEO of AB2 Bio Ltd. explained, “So we bought the rights to the drug and started working with Professor Gabay to assess its safety and efficacy in a clinical trial.”
Professor Gabay says, “Recently, we successfully demonstrated the key role of IL-18 in Still’s disease.”
Patients with the condition have a very high level of this protein, correlating with the acute phases of the disease, and this rate fluctuates according to the inflammatory activity specific to this disease.
He explained, “Our idea was therefore to block the harmful action. It is also worth pointing out that the treatments currently offered to patients are very empirical, so our goal was to finally give them a safe and approved treatment.”
While IL-18 is useful for protecting the body against external pathogens, too much of this protein leads to a harmful overactivation of the immune system, resulting in the various symptoms presented in patients.
There is a naturally occurring inhibitor (“IL-18 binding protein”), whose function is to bind to IL-18 to form an inactive complex, but people with Still’s disease produce more IL-18 than its inhibitor, which triggers their symptoms.
The main objective of this study was to verify the safety of the drug and to confirm its efficacy on the clinical manifestations of the disease.
Twenty-three patients, mostly suffering from forms of the condition that are particularly refractory to the usual treatments, were enrolled and divided into two groups, one receiving 80 mg, and the other 160 mg, via three subcutaneous injections per week for 12 weeks.
Professor Gabay summarises the results, “We were initially reassured by the safety profile: there were very few serious side effects, only one of which was possibly related to the drug itself. In addition, 50% of patients in both treatment groups showed a positive response to the drug after three weeks of treatment, and their symptoms decreased over the entire 12 weeks of follow-up.”
“In contrast, patients who didn’t have a positive 80 mg response did not have one with 160 mg either. These first results are extremely encouraging and mean we can plan a phase 3 trial to better evaluate the effectiveness of the drug.”
Researchers are currently in phase 2, which aims to ensure the safety of the product and to determine the optimal dose. Phase 3, the next step envisaged by Professor Gabay’s team together with AB2 Bio Ltd., will aim to assess the actual efficacy of the drug.
Patients with other orphan diseases associated with the dysregulated production of IL-18 and its inhibitor, for which there is no effective treatment protocol, could also benefit from these results.
In 2015, the life of a three-month-old girl was saved.
Professor Gabay tells what happened, “This child, who lives in the United States, had a rare inflammatory disease that was resistant to all treatment; she was in intensive care and was going to die. Alerted by our American colleagues, we worked with AB2 Bio Ltd. to determine what does she should be given.”
“The Food and Drug Administration granted permission to use the drug in a last-ditch attempt to halt the disease. She rapidly responded to treatment and is now 3 years old, well and can lead a normal life with IL-18 inhibitor therapy. A glimmer of hope for other patients!”