Fluoride is added to water in small concentrations by many public water suppliers to protect the teeth from decay and to build bone strength. Yet, in high concentrations (more than several parts per million) fluoride can harm human health.
According to WHO, the maximum permissible limit of fluoride in drinking water is 1.5 ppm and the highest desirable limit is 1.0 ppm. Fluoride concentrations above 1.5 ppm in drinking water cause dental fluorosis and much higher concentration skeletal fluorosis.
Defluoridation is difficult and expensive and requires a well-equipped chemical lab. Because of this, fluoride contamination in water affects a number of developing countries today and even parts of developed countries.
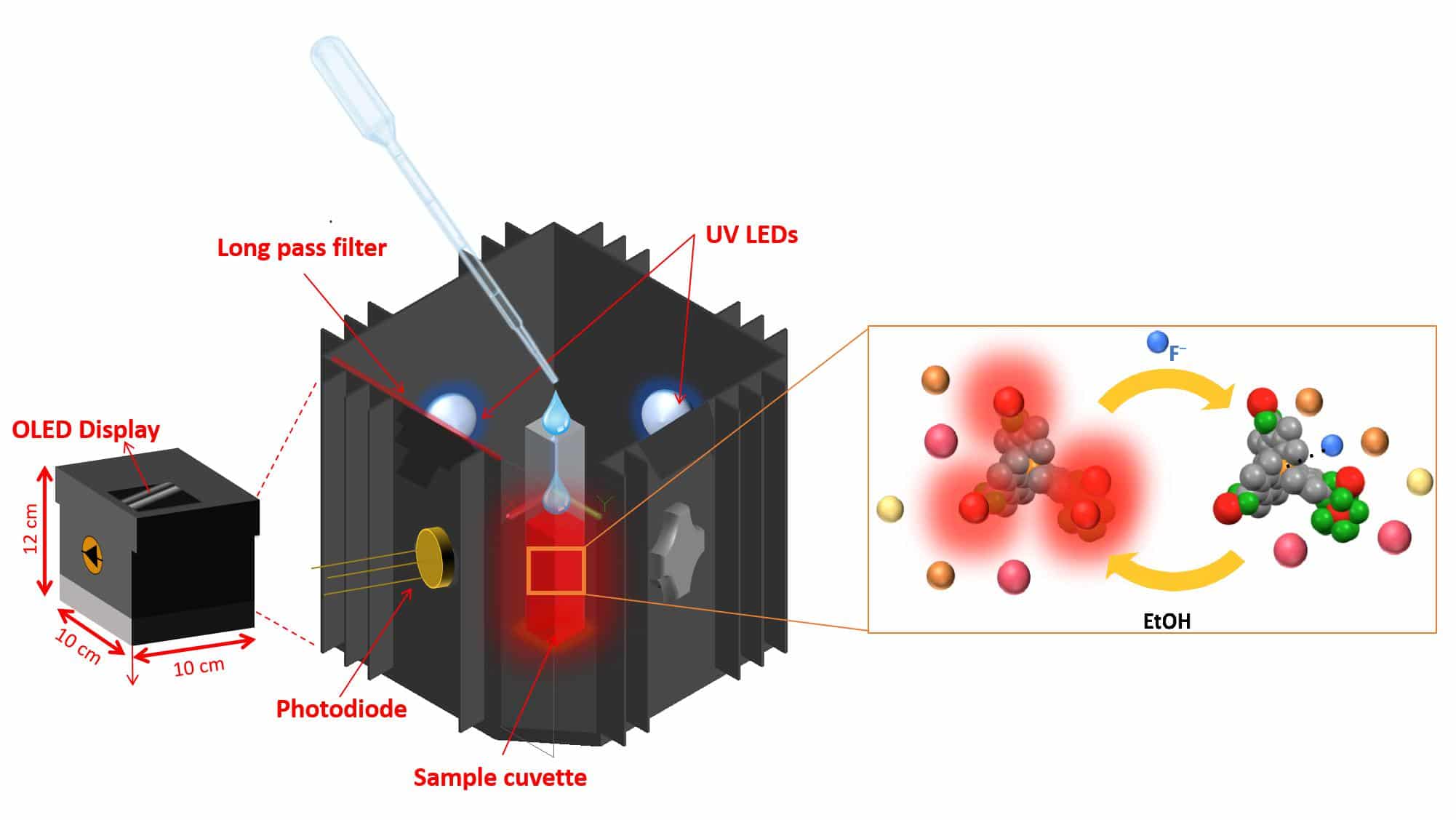
To find out the better solution for this problem, a team of scientists at the EPFL has developed a device to precisely measure fluoride concentrations using only a few drops of water- resulting in a simple change in color brightness.
Scientists dubbed this device as SION-105. It is portable, cheaper than existing methods and can be used on-site by virtually anyone.
To design this device, scientists used a material that belongs to the family of “metal-organic frameworks” (MOFs), compounds made up of a metal ion. They then connected it to organic ligands that thus form one-, two-, or three-dimensional structures.

Mish Ebrahim, the paper’s first author said, “SION-105 is luminescent by default, but darkens when it encounters fluoride ions. Add a few droplets of water and by monitoring the color change of the MOF one can say whether it is safe to drink the water or not. This can now be done on-site, without any chemical expertise.”
Scientists tested the device by determining the fluoride content in different groundwater samples from Vietnam, the United Arab Emirates, and Saudi Arabia.
Kyriakos Stylianou at the Laboratory of molecular simulation said, “This comparison showcases the performance and reliability of SION-105, which, coupled with the portability and ease-of-use of the device, make it a very user-friendly solution for water sampling in remote areas where frequent fluoride concentration monitoring is paramount.”
Having confirmed the successful function of SION-105 and the device, EPFL has now filed a patent application for it. The device was developed by a scientist at the Laboratory of molecular simulation, the Laboratory of Organometallic and Medicinal Chemistry, and the Electronic Workshop at EPFL Valais Wallis.
The study is published in the Journal of the American Chemical Society.
