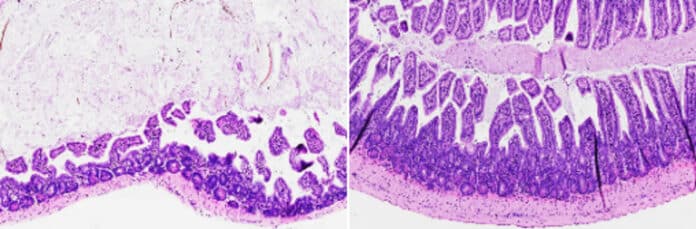
Crohn’s disease and ulcerative colitis are both examples of inflammatory bowel disease (IBD), which is characterized by an overabundance of immune cells and inflammatory signaling chemicals known as cytokines in the gut. Existing therapies only work for some people and come with various negative effects. They often suppress the immune system or specifically target specific cytokines.
For more than two decades, the Farnesoid X receptor (FXR) is known to be a master regulator protein that detects the bile acids administered to the digestive tract to aid food digestion and nutritional absorption. The body gets ready for an influx of food by turning on and off dozens of cellular programs involved in digestion, blood sugar regulation, and fat metabolism when FXR notices a change in bile acids at the start of a meal.
Fexaramine is a tablet that Salk Institute scientists created in 2015 to activate FXR in the gut. They first demonstrated that the tablet could prevent weight gain and regulate blood sugar in rats. They demonstrated in 2019 that FexD, an improved form of fexaramine, also protected changes to the gut’s stem cells caused by cancer. Their research revealed that FXR was involved in the control of inflammation.
Now, scientists have developed a drug that acts like a master reset switch in the intestines. The drug used, FexD, is found to prevent and reverse intestinal inflammation in mouse models of inflammatory bowel disease.
Senior Staff Scientist Michael Downes, co-corresponding author of the new paper, said, “Every time you eat, you’re causing small amounts of inflammation in your gut as your intestinal cells encounter new molecules. FXR makes sure inflammation stays under control during normal feeding.”
In this study, scientists found that activating FXR can ease symptoms in inflammation-driven diseases. The medicine prevented or treated intestinal inflammation in IBD-affected mice when the scientists administered oral FexD on a daily basis, either before or after the beginning of intestinal inflammation. Innate lymphoid cells are a subset of highly inflammatory immune cells, and FexD inhibited their invasion by activating FXR. As a result, levels of cytokines previously linked to IBD dropped to those typically observed in mice with normal health.
Senior Research Scientist Annette Atkins, co-author of the study, said, “When we activate FXR, we restore appropriate signaling pathways in the gut, bringing things back to a homeostatic level.”
Cytokines are partially prevented by FexD because FXR functions more like a reset switch for the immune system than an off switch. This indicates that following a dose of FexD, the immune system continues to operate normally. The molecule still needs to be refined for use in people and put through clinical trials. Still, the scientists claim their findings offer crucial insight into the intricate relationships between gut health and inflammation and may one day result in an IBD treatment.
First author Ting Fu, previously a postdoctoral fellow at Salk and now an assistant professor at the University of Wisconsin-Madison, said, “In people with IBD, our strategy could potentially be very effective at preventing flare-ups and as a long-term maintenance drug.”
Journal Reference:
- Ting Fu, Yuwenbin Li, Tae Gyu Oh, et al. FXR mediates ILC-intrinsic responses to intestinal inflammation. Proceedings of the National Academy of Sciences. DOI: 10.1073/pnas.2213041119