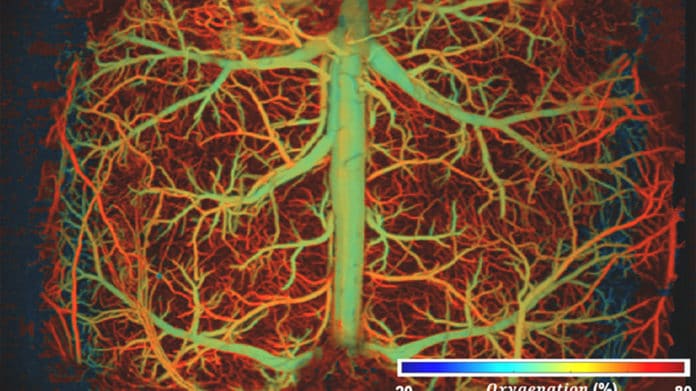
High-speed, high-resolution imaging of whole-brain hemodynamics is essential to facilitating neurovascular research. It is crucial to visualize real-time hemodynamics in complex brain vascular networks and track fast pathophysiological activities at the microvessel level.
Doing these things individually is possible, but it isn’t easy to do them together. A new study solved this long-standing trade-off.
Biomedical engineers at Duke University have developed a method to scan and image brain activity in real time with enough resolution. The technique breaks long-standing speed and resolution barriers in brain imaging technologies. As engineers reported, the method could unveil new insights into neurovascular diseases like stroke, dementia, and even acute brain injury.
The new method- ultrafast photoacoustic microscopy, or UFF-PAM- relies on hardware advancements and machine learning algorithms to upgrade the technique.
Photoacoustic microscopy captures comprehensive images of organs, tissues, and cells throughout the body by combining the properties of light and sound. A laser is used to direct sunlight into a specific tissue or cell. When the laser strikes the cell, it instantly heats up and expands, producing an ultrasonic signal that goes back to the sensor.
Junjie Yao, an assistant professor of biomedical engineering at Duke, said, “The resulting images looked as detailed as the high-resolution images we would usually get if we went at a much slower speed. We didn’t need to sacrifice a full field of view.”
On the hardware side of UFF-PAM, a polygon scanning system sends more laser bursts to a larger area. In contrast, a new scanning mechanism allows the laser scanner and ultrasound sensor to operate simultaneously.
Junjie Yao, an assistant professor of biomedical engineering at Duke, said, “These changes doubled the speed of their device, making UFF-PAM the fastest imaging technology in the photoacoustic community.”
Engineers then developed a machine learning algorithm that improved the resolution of their images. They used over 400 photos of mouse brains acquired in earlier investigations to train them to recognize vasculature in the brain. Even though each brain is unique, the algorithm learned to recognize common structures and used this information to fill in previously blank pixels.
Yao said, “The resulting images looked as detailed as the high-resolution images we would usually get if we went at a much slower speed, and we didn’t need to sacrifice a full field of view.”
Using their method, scientists visualized how blood vessels in a mouse’s brain responded to hypoxia, drug-induced hypotension, and ischemic stroke. During the hypoxia challenge, UFF-PAM tracked how oxygen moved through the brain and showed that low oxygen levels caused blood vessels to dilate.
In the second challenge, the team used the drug sodium nitroprusside (SNP), which is commonly used to treat high blood pressure. Previously, scientists thought that SNP causes all the blood vessels in the brain to dilate. But Yao and his team instead showed that only the larger blood vessels open up while smaller blood vessels constrict.
Yao said, “Because we quickly got a high-resolution view of the smaller vessels, we saw that dilation is not the universal response to the drug. We saw that these small vessels couldn’t provide enough oxygen and nutrients to the tissue, which caused damage.”
Scientists observed how the brain responds to stroke and starts recovering in the final stage. They observed that the blood vessels in the affected area constrict immediately after stroke, compressing neighboring vessels in a phenomenon called a spreading depolarization wave.
Because of the large field of view and high imaging speed, the team pinpointed the wave’s starting position and tracked its movement as it propagated throughout the brain.
In the future, scientists plan to use UFF-PAM to explore additional brain disease models, like dementia, Alzheimer’s disease, or even Long COVID. They also plan to expand the tool’s use outside the brain to image organs like the heart, liver, and placenta.
Journal Reference:
- Xiaoyi Zhou, Qiang Huang, et al. Real-time whole-brain imaging of hemodynamics and oxygenation at micro-vessel resolution with ultrafast wide-field photoacoustic microscopy. DOI: 10.1038/s41377-022-00836-2