Researchers at McGill University have discovered a new technique for measuring how quickly medications interact with their molecular targets. The discovery provides scientists a new way to investigate the effectiveness of medication candidates that might otherwise have been overlooked.
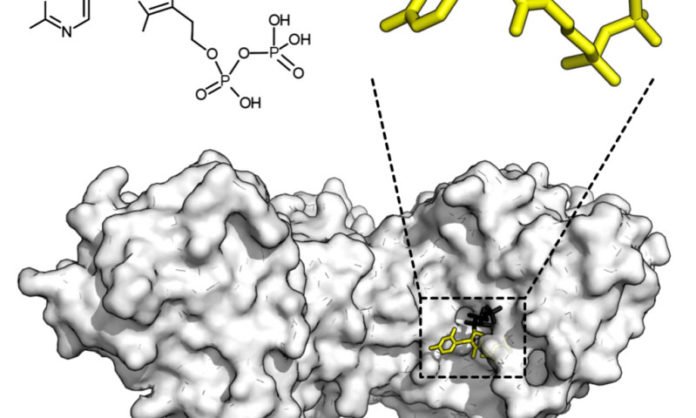
This new method focuses on the principle of enzyme inhibition. Countless pharmaceuticals, ranging from antibiotics to chemotherapy medications, work by blocking the action of enzymes, and the search for new enzyme-inhibiting substances remains a major focus of medication development.
The McGill team, led by chemistry professors Nicolas Moitessier and Anthony Mittermaier, demonstrate the use of isothermal titration calorimetry (ITC) to gauge the heat generated by enzyme activity and thereby the rates at which inhibitor substances blocked that activity.
Anthony Mittermaier explained, “One key difference between ITC and other methods is that ITC measures the rate of reaction directly.”
Existing methods for measuring enzyme activity look at that activity indirectly, by measuring changes in concentration caused by enzymatic catalysis as a function of time. These measurements often depend on special reagents that change color or fluorescence when acted on by the enzyme and require a unique test to be developed for each enzyme being studied.
Because ITC measures the production of heat, a near-universal feature of chemical reactions, it can be applied to just about any enzyme.
Mittermaier said, “ITC is as close as you can get to a universal enzyme test.”
In addition to its generality, the ITC method gives a direct read-out of enzyme activity because it detects heat flow in real time. By providing a direct window on the reaction, ITC offers researchers a better insight into the mechanisms by which enzyme inhibition proceeds.
It is usually very challenging, and sometimes next to impossible, to obtain this information from conventional assays.
The real-time nature of ITC is exceptionally favorable for researchers investigating covalent inhibitors. These strongly binding molecules have potential as long-acting drugs but had previously fallen out of favor in drug development due to toxicity concerns.
The insight ITC offers into the relationship amid an inhibitor’s molecular structure and how it reacts with its target will support renewed interest in covalent inhibitors and facilitate the work of developing them into drugs that are both highly effective and safe.
The research published online today in the journal Nature Communications.