As I told you previously, hydrogen is considered as a future energy resource. Although, water is a traditional source of hydrogen for a fuel cell, which splits into hydrogen and oxygen. But oxygen is a low-value product. But have you ever thought, nail polish is developed while stimulating fuel cells?
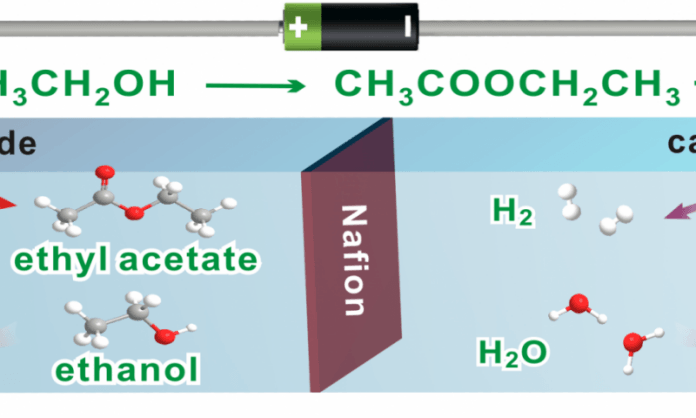
Scientists have developed a novel approach which can generate not only hydrogen but also important chemicals like the most usual ingredient in nail polish. They found a new way to make ethyl acetate while generating H2 gas to power fuel cells.
Scientists just recognized, it is essential to have a source that consists of hydrogen and oxygen for generating hydrogen. However, water is the conventional source, other substances could also fit the bill. Scientists developed various natural molecules to use in consumer products by selecting the right initial ingredient and catalyst.
For being natural as much as it possible, scientists designed a catalyst called cobalt and oxygen. After running the water-splitting reaction with ethanol and their cobalt oxide catalyst, they produced H2 gas and ethyl acetate. Ethyl acetate a chemical used to make nail polish, clean circuit boards and decaffeinate coffee and tea. Additionally, ethyl acetate was the only product of the reaction aside from H2. Thus, scientists would be able to obtain pure ethyl acetate without energy-consuming purification processes.
According to scientists, this advance could pave the way for completely green systems that generate Hydrogen. Additionally, it could be used a range of natural chemicals at room temperature from renewable electrical sources.