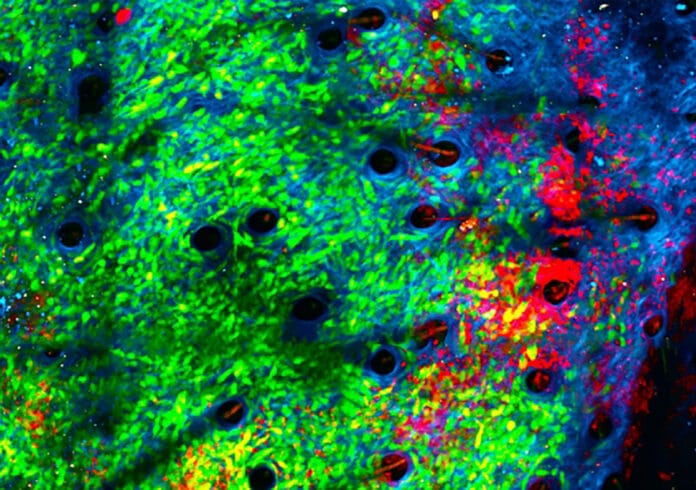
The inflammatory microenvironment of solid tumors creates a pro-tumorigenic milieu that resembles chronic inflammation akin to a subverted wound healing response. A new study investigated the effect of converting the tumor microenvironment from a chronically inflamed state to one of acute microbial inflammation by injecting microbial bioparticles directly into tumors.
The study, conducted by UNSW Sydney and the Garvan Institute of Medical Research, found that the primary responder cells of the immune system are triggered to fight rather than defend a tumor when bacteria are introduced to the microenvironment of a tumor.
The research team found that the protective role of neutrophils is reversed when inactivated samples of the Staphylococcus aureus bacterium are injected into the tumor microenvironment or the region surrounding the tumor.
According to scientists, the findings could lead to better treatments to improve outcomes for people with advanced or previously untreatable cancers. It also helped advance our understanding of acute inflammation to advance microbial therapy for cancer.
Associate Professor Tatyana Chtanova at UNSW’s School of Biotechnology and Biomolecular Sciences and Head of the Innate and Tumour Immunology Lab at Garvan said, “In our study, we sought to develop new immunotherapies that use different modes of action that could complement and enhance existing immunotherapies.”
“We show how acute inflammation can be harnessed to achieve ongoing anti-tumor function in immune cells. We also show how microbial therapy can be successfully combined with an existing type of therapy, known as checkpoint inhibitor therapy, to amplify anti-cancer capabilities.”
In their study, scientists decided to use a different type of immunotherapy that targets neutrophils. They wanted to get insights into how generating acute inflammation in the immunosuppressive tumor microenvironment affects outcomes.
Using an innovative imaging technique known as intravital imaging, the team observed the tumors in real-time.
A/Prof Chtanova said, “Since attacking bacteria is the reason for neutrophils’ existence, we had a good clue that introducing bacteria would bring neutrophils to the site and activate them. We discovered it’s very effective in getting them to kill the tumors, chewing up their matrix.”
The study also discovered that neutrophils start secreting chemicals that will draw additional fighting T cells when exposed to germs.
The study’s first author, Dr. Andrew Yam, clinical medical oncologist at The Kinghorn Cancer Centre and Ph.D. student at Garvan, said, “We’ve shown that microbial therapy is an effective booster for checkpoint inhibitor therapy. We hope this synergistic effect will ultimately lead to better treatments to improve outcomes for patients with advanced or previously untreatable cancers.”
A/Prof Chtanova said, “This study focused on primary tumors, the first tumor in the body. So far, we have shown that our microbial therapy can inhibit the growth of primary tumors and protect against tumor recurrence, which is a major clinical challenge. This suggests that our microbial therapy is achieving not just short-term and localized, but long-lasting and systemic anti-tumor immunity.”
“Our next step is to extend these findings to develop a pathway to treat cancers that have metastasized to different locations.”
Journal Reference:
- Andrew Yam, Jacqueline Bailey, et al. Neutrophil conversion to a tumor-killing phenotype underpins effective microbial therapy. Cancer Research. DOI: 10.1158/0008-5472.CAN-21-4025