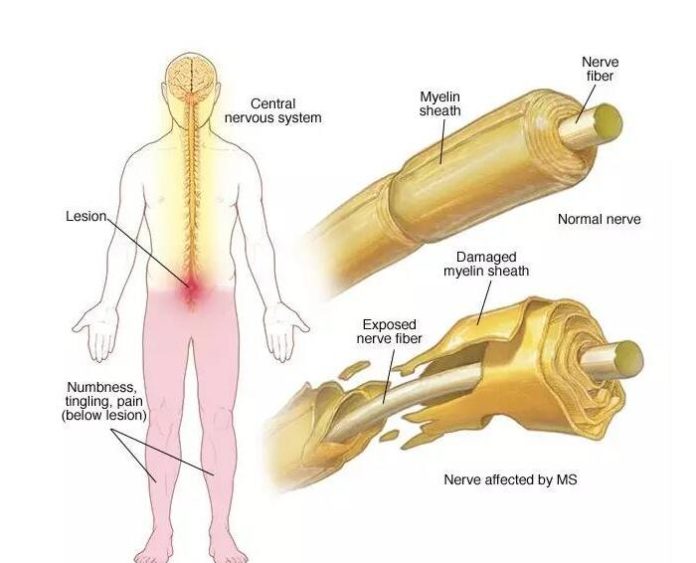
The misfortune or harm of myelin, a cell sheath that encompasses and protects nerves, is the sign of the insusceptible intervened neurological confusion different sclerosis (MS). At the point when sections of this defensive film are harmed, nerve motivations can be disturbed. Indications extend from shivering and deadness to shortcoming, torment, and loss of motion.
There is as of now no solid method to straightforwardly picture demyelination. Doctors depend on magnetic resonance imaging (MRI), however, regardless of high determination pictures, MRI isn’t quantitative and can’t recognize demyelination and irritation, which regularly exist together in individuals with MS.
On January 12, 2017, online issue of the journal Scientific Reports, a multi-institutional group based basically at the University of Chicago Medicine and the National Institutes of Health, portray early trial of a novel negligibly obtrusive approach to evaluate myelin harm utilizing positron discharge tomography (PET).
These PET outputs utilize a radioactive atom intended to target voltage-gated potassium channels, a protein found on demyelinated axons. The PET pictures, in view of the discovery of this particle, give quantitative data about fundamental biochemical procedures.
Study author Brian Popko said, “In healthy myelinated neurons, potassium channels are usually buried underneath the myelin sheath. When there is a loss of myelin, these channels become exposed. They migrate throughout the demyelinated segment and their levels increase.”
These uncovered neurons release intracellular potassium. This abandons them unfit to proliferate electrical motivations, which causes a portion of the neurological manifestations found in MS. So we built up a PET tracer that can target potassium channels,” Popko said.
The group began with a current MS sedate, 4-aminopyridine (a.k.a. dalfampridine), which can tie to uncovered potassium channels. This can in part reestablish nerve conduction and lighten neurological side effects in MS patients. Utilizing mouse models of MS, incorporating some created in the Popko lab, the scientists demonstrated that the medication aggregated in the demyelinated, or revealed, territories of the focal sensory system.
At that point, with assistance from partner Pancho Bezanilla, Ph.D., educator of organic chemistry and sub-atomic science at the University of Chicago, the group analyzed a few fluorine-containing subsidiaries of 4-aminopyridine for authoritative to K+ stations. They found that 3-fluoro-4-aminopyridine (3F4AP) has the coveted properties, so they marked the particle with fluorine-18, which is effectively recognized by PET.
Pedro Brugarolas, Ph.D., first author of the paper said, “All existing PET tracers used for imaging demyelination bind to myelin and, consequently, demyelinated lesions show as decreases in the signal, which can be problematic for imaging small lesions. 3F4AP is the first tracer whose signal increases with demyelination, potentially solving some of the problems of its predecessors.”
“A tracer to monitor changes in something as ubiquitous as potassium channels could have applications for other diseases where these channels are involved.”
In collaboration with scientists at the NIH, the researchers conducted a study in healthy monkeys. They confirmed that radiolabeled 3F4AP enters the brain of primates and localizes to areas where there is little myelin.
That list includes leukodystrophies, traumatic brain injury, spinal cord injury and “even maladies not traditionally associated with demyelination. For example, ischemia, psychiatric disorders, and neurodegenerative diseases, including Alzheimer’s.