Gadolinium contrast medium is used in about 1 in 3 MRI scans to improve the clarity of the images or pictures of your body’s internal structures. This improves the diagnostic accuracy of the MRI scan.
A new study by Rice University engineers suggests that the Gadolinium-based contrast agents in MRI could be improved.
Engineers studied the use of nuclear magnetic resonance tools in the oil industry to characterize deposits underground. They suggest that molecular dynamics simulations could be used to optimize these tools.
Philip Singer of the George R. Brown School of Engineering said, “There are roughly 100 million MRIs taken worldwide every year, and about 40% of them use gadolinium-based contrast agents, but the way they model MRI response to these agents hasn’t changed significantly since the 1980s. We thought it would be a good testbed for our ideas.”
The team demonstrated that limiting several parameters in simulations could potentially improve the analysis of gadolinium-based contrast agents. They also tested the effectiveness of simulations at imaging for clinical diagnosis.
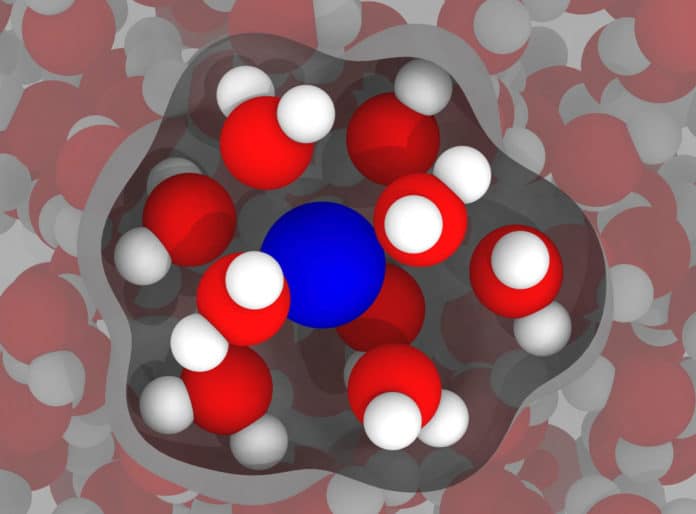
Dilip Asthagiri of the George R. Brown School of Engineering said, “Doctors use MRI devices to “see” the state of soft tissues inside the body, including the brain, by inducing magnetic moments in the hydrogen nuclei of ever-present water molecules to align along the magnetic field. The device detects bright spots when the aligned nuclei “relax” back to thermal equilibrium following an excitation, and the faster they relax, the brighter the contrast.”
“That’s where paramagnetic gadolinium-based contrast agents come in. Gadolinium ions increase sensitivity and make the signal brighter by decreasing the T1 relaxation time of hydrogen nuclei. Our ultimate goal is to help the optimization and design of these agents.”
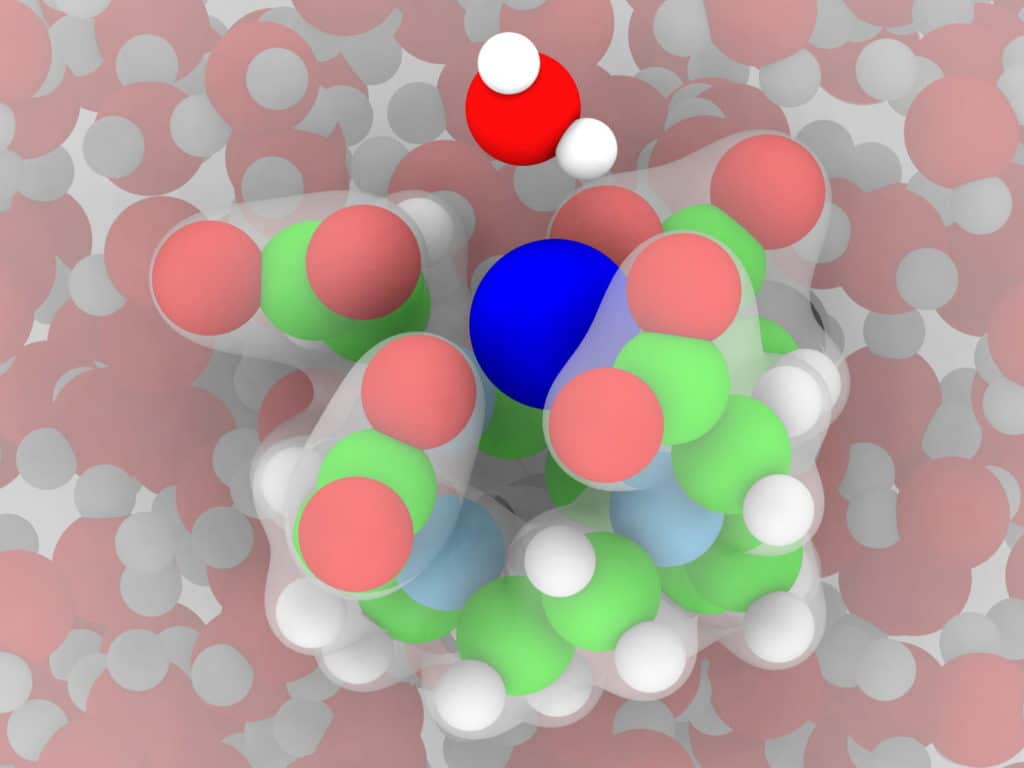
Singer said, “Typically, gadolinium is “chelated” (claws) —surrounded by metal ions—to make it less toxic. The body doesn’t remove gadolinium by itself and needs to be chelated so the kidneys can get rid of it after a scan. But chelation also slows the molecular rotation, and that creates better contrast in the MRI image.”
“In this case, these claws grip the gadolinium to make it stable. We hope our models help us design a stronger grip, which will make them safer while maximizing their ability to increase contrast.”
Asthagiri said, “We are also adapting its models beyond interactions with water. In biological systems, cells have other constituents like osmolytes and denaturants like urea, so we’re modeling gadolinium with these different environments to build toward a variety of applications.”
Journal Reference:
- Philip M. Singer et al., Predicting 1H NMR relaxation in Gd3+-aqua using molecular dynamics simulations, Physical Chemistry Chemical Physics (2021). DOI: 10.1039/D1CP03356E