Atoms that makeup everything- bond together by sharing electrons. They can be considered as a glue of life. Nagoya University applied physicist Yoshihiko Okamoto and colleagues have found a highly unusual version of this glue: a regular triangular molecule was formed of three atoms bonded together by two electrons.
Scientists have detected this highly unusual atomic configuration in a tungsten-based material.
Until now, such type of bond has been detected in the trihydrogen ions found in interstellar material. This discovery of so-called tritungsten molecules in single crystals of caesium tungsten oxide (CsW2O6) could reveal compounds with interesting electronic properties.
CsW2O6 conducts electricity at room temperature but changes into an insulating material when it is cooled below -58°C. Scientists found it challenging to study how the atomic structure of this type of material changes in response to temperature.
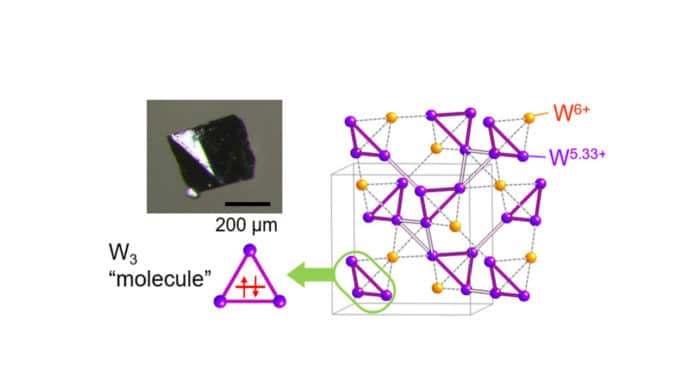
Scientists found a solution to this: They synthesized very pure single crystals of CsW2O6 and bombarded them with X-ray beams at room temperature and -58°C. The tungsten molecules in the conducting crystal form three-dimensional networks of tetrahedral pyramids connected at their corners, known as a pyrochlore structure. The bonds between the molecules form due to a symmetrical sharing of electrons between them.
Be that as it may, when the compound is cooled, the electrons re-arrange and two types of tungsten atoms forms within the tetrahedra, each with an alternate ‘valence’, or bonding power with different atoms. This, thus, mutilates the lengths of tungsten bonds with oxygen atoms in the compound, prompting an increasingly compacted shape.
Critically, the tungsten atoms with lower valence form small and large triangles on the sides of the tungsten tetrahedra, with the exceptionally unusual tritungsten molecules forming on the small triangles. The three tungsten atoms forming the points of these triangles share only two electrons between them to keep them bonded together.
Nagoya University applied physicist Yoshihiko Okamoto said, “To our knowledge, CsW2O6 is the only example where this type of bond formation, where several atoms share only a few electrons, appears as a phase transition.”
Further plans include investigating compounds with pyrochlore structures.
Journal Reference:
- Okamoto, Y., Amano, H., Katayama, N. et al. Regular-triangle trimer and charge order preserving the Anderson condition in the pyrochlore structure of CsW2O6. Nat Commun 11, 3144 (2020). DOI: 10.1038/s41467-020-16873-7