In a new study by the University of Oxford (United Kingdom) and University of Jyväskylä (Finland), scientists found a totally new method of reactivity for compounds of aluminum – a standout amongst the most innovatively imperative components of the Periodic Table. This new capacity may discover additionally use in applications including the arrangement of new metal-carbon and metal-metal bonds.
Trivalent aluminum compounds– as each science understudy will tell – are electron lacking and go about as Lewis acids (electron acceptors) in their concoction responses. Various manufactured changes and broadly utilized modern procedures (e.g. the creation of polythene) draw on this trademark concoction property.
Aluminum compounds in the +1 oxidation state are additionally known, obviously, however, the turn around extremity ‘umpolung’ procedure of utilizing an aluminum reagent as the nucleophilic accomplice in Al-E bond-framing substitution responses (E = H, C and so forth.) has not been tentatively open.
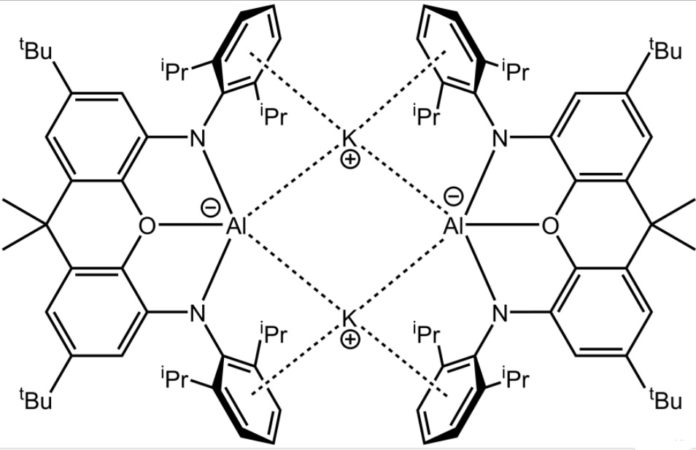
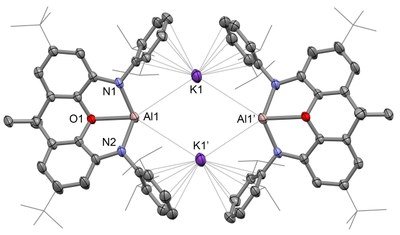
This is for the first time, scientists synthesized anionic aluminum (I) at room temperature. The molecules act as nucleophiles. A dimethylxanthene-settled potassium aluminyl compound can be integrated by potassium lessening of the relating iodide, and presentations phenomenal reactivity: (I) in ‘umpolung’ mold as an aluminum-focused nucleophile for the arrangement of Al-E covalent securities (E = H, C) by means of substitution science; and (ii) in the C-H oxidative expansion of benzene at a solitary very much characterized primary gathering metal focus.
Scientists believe that these outcomes portray a stage change in the science available to a standout amongst the most mechanically critical components in the Periodic Table – allegorically, it turns aluminum science on its head.
The research was published in Springer Nature on the 16th of April 2018.