Determining different stages of a chemical reaction is quite tricky. The formation of bonds is central to our understanding of all chemical processes.
In a new study, scientists from Nottingham Trent University, the University of Southampton, and the University of Warwick have successfully monitored the different stages of a chemical reaction.
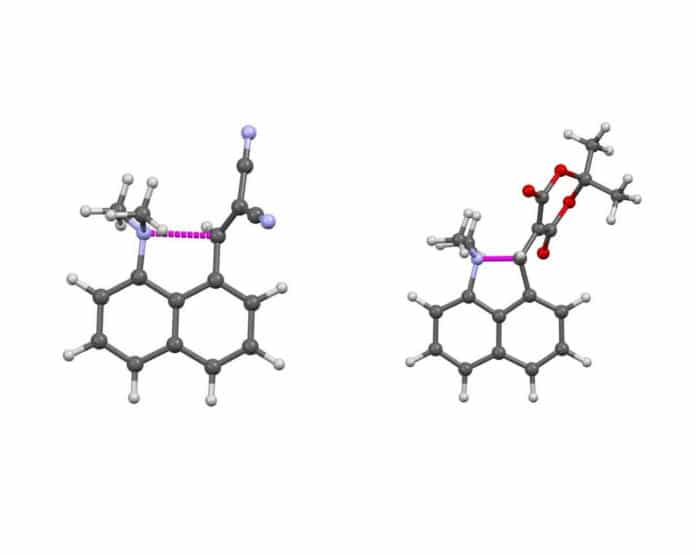
Using high-quality X-Ray diffraction data and advanced solid-state nuclear magnetic resonance (NMR) techniques, scientists could trap the stages in a crystalline state. They measured and observed the degree of bond formation, both in terms of the increasing participation of electrons and the magnetic interaction between the two atoms at either end of the bond, as the bond forms.
Scientists studied a series of six molecules in which a bond between a nitrogen and carbon atom was partially formed to different degrees. They followed the redistribution of electrons on bond formation.
Using the NMR technique, they monitored the magnetic interaction between the two atoms after forming the bond.
Lead researcher John Wallis, Emeritus Professor in Nottingham Trent University’s School of Science and Technology, said, “Our work provides the methods for studies on other bond-forming processes. This is important because catalysts aim to speed up reactions by stabilizing the pathway by which bonds are formed and broken.”
Journal Reference:
- Dr. Gregory, J. Rees et al. Mapping of N−C Bond Formation from a Series of Crystalline Peri-Substituted Naphthalenes by Charge Density and Solid-State NMR Methodologies. DOI: 10.1002/anie.202111100