In a crystal, All atoms shape an intermittent structure called a lattice, where the molecules are arranged in a tedious example in three dimensions. The properties of electrons living in this space commonly comply with a similar periodicity.
In a new study, researchers from the U.S. Department of Energy’s (DOE) Argonne National Laboratory have determined that electrons in some oxides can experience an “unconventional slowing down” of their response to a light pulse.
The researchers describe the behavior as lasting about a millionth of a second, which is still a million times slower than traditional electronic recovery times.
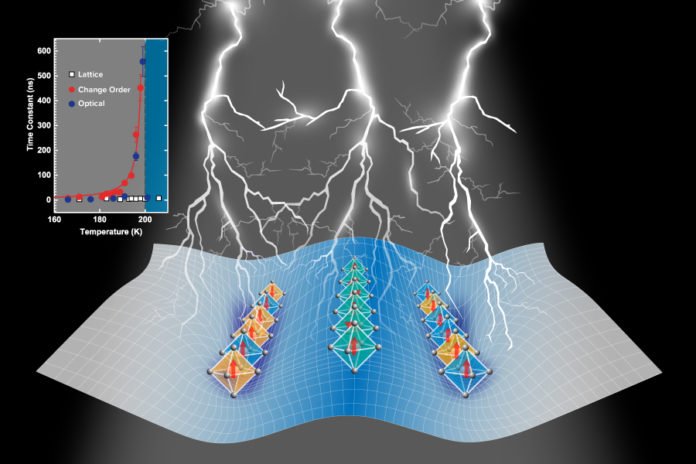
During the study, scientists studied the material known as La1/3Sr2/3FeO3 and found that electrons can be very slow to return to their homes after being kicked out from the ‘ordered states’.
In any case, underneath a temperature of about minus 100 degrees Fahrenheit, the electrons in the investigation material, lanthanum strontium ferrite, find it more vigorously worthwhile to coordinate with the lattice and magnetism on the iron particles, to frame another periodic structure called an attractively determined, charge-requested state.
The behavior happens near a temperature that denotes a stage progress — like the manner by which 32 degrees Fahrenheit denotes the stage change from water to ice. In any case, the phase transition examined here is unconventional in light of the fact that it denotes a progress between a magnetic insulator and a non-magnetic metal.
Since the electrons are lighter than the atoms in the lattice, they, for the most part, respond all the more rapidly to the light pulse, subsiding into their new state before the lattice stops. However, for this situation, the electronic recuperation endures any longer than the lattice recuperation.
The researchers conducted transient optical reflectivity measurements at CNM with Argonne scientist Richard Schaller and observed that, as the temperature approaches the phase transition, the electron relaxation slows down by orders of magnitude. Complimentary hard X-ray diffraction at the APS measured structural changes to determine how quickly the lattice and charge-ordered phase evolved.
Scientists noted that the strange conduct of electrons is likely a consequence of magnetic interactions. Routinely, the two areas on either side of a first-arrange stage change are very particular, similar to water and ice.
They found that, due to magnetic interactions close to the phase transition, there are actually two different kinds of charge-ordered states. When the excited electrons try to go back to the charge-ordered phase, they find a very shallow downhill slope into one of the two charge-ordered states, which ultimately slows down the ordering processes.
For both Anand Bhattacharya, an Argonne materials scientist and Haidan Wen, Argonne physicist, understanding these “slow” procedures in the realm of ultrafast phenomena may offer better approaches to settle or increment the lifetime of outlandish transient expresses that happen after a material is hit with a light pulse. Such bits of knowledge may uncover conceivably valuable properties identified with magnetism, conductivity or even superconductivity.
Other co-authors are from the University of Illinois at Chicago, Northwestern University, and Dublin City University.
The study is published in Nature Communications.