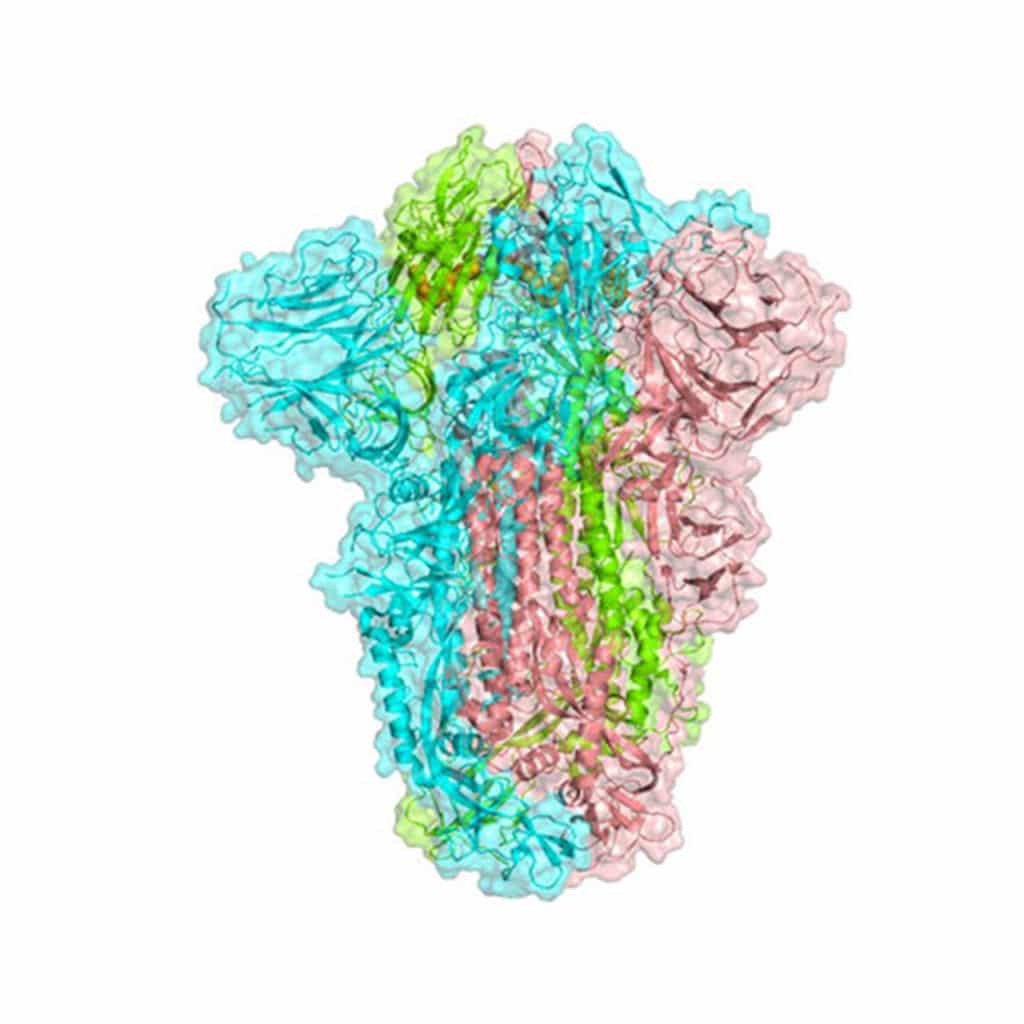
COVID-19, caused by severe acute respiratory syndrome-coronavirus-2 (SARS-CoV-2), represents a global crisis. Coronavirus entry into host cells is mediated by the transmembrane spike (S) glycoprotein that forms homotrimers protruding from the viral surface.
This spike protein binds to the cell surface, enabling the virus to enter into the cells and replicating, causing widespread damage.
Using a powerful imaging technique, electron cryo-microscopy (cryo-EM), scientists from the University of Bristol made a groundbreaking discovery. They analyzed SARS-CoV-2 Spike at near-atomic resolution and identified its molecular composition.
What they found is quite surprising. They found the presence of a small molecule, linoleic acid (LA), buried in a tailor-made pocket within the Spike protein.
LA is a free fatty acid, which is indispensable for many cellular functions. The human body cannot produce LA. Instead, the body absorbs this essential molecule through diet. Intriguingly, LA plays a vital role in inflammation and immune modulation, both key elements of COVID-19 disease progression.
This discovery offers a first direct link between LA, COVID-19 pathological manifestations, and the virus itself.
Professor Imre Berger from the Max Planck-Bristol Centre for Minimal Biology said, “We were truly puzzled by our discovery and its implications. Here we have LA, a molecule at the center of those functions that go haywire in COVID-19 patients, with terrible consequences. And the virus that is causing all this chaos, according to our data, grabs and holds on to exactly this molecule – basically disarming much of the body’s defenses.”
Professor Christiane Schaffitzel from Bristol’s School of Biochemistry said, “From other diseases, we know that tinkering with LA metabolic pathways can trigger systemic inflammation, acute respiratory distress syndrome, and pneumonia. These pathologies are all observed in patients suffering from severe COVID-19. A recent study of COVID-19 patients showed markedly reduced LA levels in their sera.”
“COVID-19 continues to cause widespread devastation, and in the absence of a proven vaccine, it is vital that we also look at other ways to combat the disease. If we look at HIV, after 30 years of research, what worked, in the end, is a cocktail of small-molecule antiviral drugs that keep the virus at bay. Our discovery of a druggable pocket within the SARS-CoV-2 Spike protein could lead to new antiviral drugs to shut down and eliminate the virus before it entered human cells, stopping it firmly in its tracks.”
Enabled by Oracle high-performance cloud computing, a 3D structure of SARS CoV-2 Spike protein was generated, allowing the researchers to peer deep inside the Spike identifying its molecular composition.
Alison Derbenwick Miller, Vice President, Oracle for Research, added: “Oracle for Research unites researchers and cloud computing to help bring about beneficial change for our planet and its people. SARS-CoV-2 and COVID-19 cause global devastation, and research efforts to find vaccines and treatments cannot move quickly enough. We are so pleased that Oracle’s high-performance cloud infrastructure enabled Professors Berger and Schaffitzel to examine the molecular structures of the coronavirus spike protein and make this powerful and unexpected discovery that could help curb the pandemic and save lives.”
The team included Bristol UNCOVER Group, Bristol biotech Imophoron Ltd, the Max Planck Institute for Biomedical Research in Heidelberg, Germany, and Geneva Biotech Sàrl, Switzerland.
Journal Reference:
- C Toelzer et al. Free fatty acid-binding pocket in the locked structure of SARS-CoV-2 spike protein. DOI: 10.1126/science.abd3255