Silver nanoparticles measuring between one and 100 nanometers (billionth of a meter) in size are being incorporated outside the United States into a variety of kitchen products in the form of food contact materials (FCMs).
In any case, the utilization of these things raises worries that the nanoparticles in them will relocate into food and nature, and thusly, regardless of whether this poses danger to human health.
In order to overcome this issue, government bodies around the world have published guidance documents, set policies and considered regulations. Now in a new study, scientists have portrayed how they reenacted cut movement, washing and scratching on nanosilver-containing slicing sheets to check whether buyer utilize hones influence nanoparticle discharge.
Scientists developed a test method and used in five different scenarios each mimicking an alternate sort and level of wear ordinarily observed with human utilize. In each test, they moved three grating surfaces forward and backward crosswise over examples of nanosilver-empowered cutting board material.
Though the method is expected to help regulatory bodies identify if any safety or health risks exist from silver nanoparticles in food contact materials and if so, find ways to deal with them appropriately before they are approved for sale in the United States.
NIST physical scientist Keana C.K. Scott said, “A custom-designed razor blade replicated knife cuts, a piece of scrubbing pad mimicked normal dishwashing conditions and a tungsten carbide burr imitated scratching by metal utensils. The washing and scratching scenarios were done at one or two levels of abrasion; for example, 500 and 5,000 cycles for the scratching simulation.”
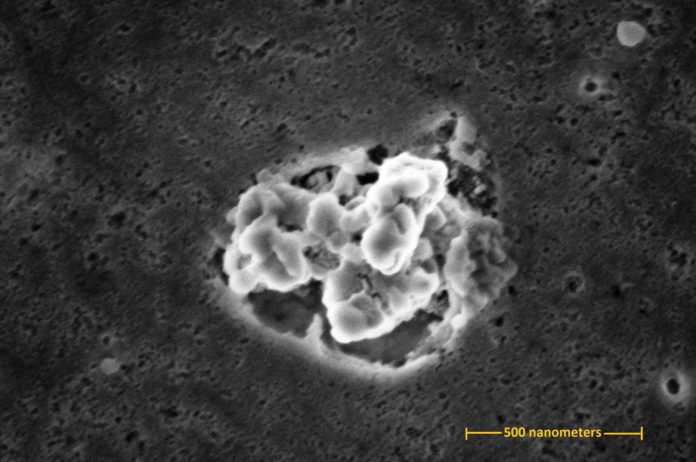
After the test, scientists used sticky tape to check whether free silver nanoparticles were available and could be expelled from the well used cutting-board tests. Scanning electron microscopy (SEM) at NIST and inductively coupled plasma mass spectrometry (an unbelievably delicate strategy for distinguishing metal particles) at the FDA demonstrated that bits of cutting board polymer were discharged by scraped area and that a portion of these contained implanted silver. Be that as it may, free silver nanoparticles were not found on the SEM-examined tape.
Scientists also determined that how much, if any, silver ions and intact silver nanoparticles would migrate away from cutting boards when exposed to water and acetic acid. They found that the concentrations of ionic and particulate silver found in both solutions were very low.
There was no discernable difference in the silver migration observed from the new and unused nanosilver-enabled cutting boards compared with the ones that were cut, washed or scratched.
NIST research chemist David Goodwin, another author on the paper said, “Now that we’ve shown that the migration evaluation method works, it can be used to help answer this and other questions about what happens when people use FCMs with nanoparticles. In turn, those findings should be valuable for the bodies that must determine any health or safety risks.”
The study was conducted by the National Institute of Standards and Technology (NIST) and the U.S. Consumer Product Safety Commission (CPSC).
The paper is published in Food Additives and Contaminants.