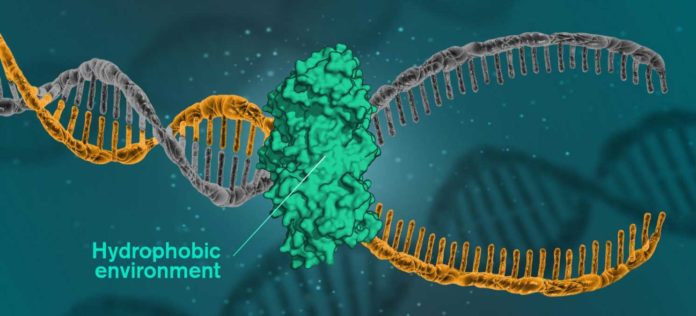
DNA is developed of two strands, comprising of sugar molecules and phosphate groups. Between these two strands are nitrogen bases, the compounds which make up organisms’ genes, with hydrogen bonds between them. Until now, it was regularly believed that those hydrogen bonds were what held the two strands together.
But now, scientists at the Chalmers University of Technology have shown that the key to DNA’s helical structure might be that the molecules have a hydrophobic inside in an environment comprising mainly of water. The discovery holds the potential to be used for research in medicine and life sciences.
Due to the hydrophobic interior, the DNA molecules’ nitrogen bases are indeed hydrophobic, pushing away the surrounding water. When hydrophobic units are in a hydrophilic environment, the group minimizes their exposure to the water.
The job of the hydrogen bonds, which were recently observed as critical to holding DNA helixes together, appears to be more to do with sorting the base pairs so they connect in the correct sequence. The disclosure is urgent for understanding DNA’s association with its environment.
Bobo Feng, one of the researchers behind the study, said, “Cells want to protect their DNA and not expose it to hydrophobic environments, which can sometimes contain harmful molecules. But at the same time, the cells’ DNA needs to open up to be used.”
“We believe that the cell keeps its DNA in a water solution most of the time, but as soon as a cell wants to do something with its DNA, like read, copy or repair it, it exposes the DNA to a hydrophobic environment.”
With regards to fixing damaged DNA, the damaged regions are exposed to a hydrophobic environment to be replaced. A catalytic protein makes the hydrophobic environment. This kind of protein is key to all DNA repair, which could be the way to battling numerous sicknesses.
Understanding these proteins could yield numerous new insights into how we could, for instance, battle resistant bacteria or possibly even cure cancer.
Bacteria use a protein called RecA to repair their DNA, and the specialists accept their outcomes could provide new knowledge into how this procedure works – conceivably offering strategies for halting it and subsequently killing the bacteria.
Bobo Feng said, “To understand cancer, we need to understand how DNA repairs. To understand that, we first need to understand DNA itself. So far, we have not, because we believed that hydrogen bonds were what held it together.”
Scientists experimented in the lab to study how DNA behaves in a hydrophobic environment. They used a hydrophobic solution of polyethylene glycol and, step-by-step, changed the DNA’s surroundings from a naturally hydrophilic environment to a hydrophobic one.
Through this experiment, they wanted to know if there is a limit to where DNA starts to lose its structure when it does not have a reason to bind because the environment is no longer hydrophilic.
Scientists found that the DNA molecules’ characteristic spiral form started to unravel when the solution reached the borderline between hydrophilic and hydrophobic.
Upon closer inspection, they observed that when the base pairs split from one another (due to external influence or directly from random movements), holes are formed in the structure, allowing water to leak in. Because DNA wants to keep its interior dry, it presses together, with the base pairs coming together again to squeeze out the water. In a hydrophobic environment, this water is missing, so the holes stay in place.
Bobo Feng said, “Now, we have shown that instead, it is the hydrophobic forces that lie behind it. We have also shown that DNA behaves differently in a hydrophobic environment. This could help us to understand DNA and how it repairs. Nobody has previously placed DNA in a hydrophobic environment like this and studied how it behaves, so it’s unsurprising that nobody has discovered this until now.”
The study is published in Proceedings of the National Academy of Sciences of the United States of America (PNAS).